Yulia A Belaya,1 Olga F Belaya,2 Vladimir G Petrukhin,1 Svetlana M Bystrova,1 Maria S Vakhrameeva,1 Alexander V Pronin1
1 Federal State Budgetary Institution «National Research Center for Epidemiology and Microbiology named after the Honorable Academician NF Gamalei» of the Ministry of Health of the Russian Federation
2 Federal State Autonomous Educational Institution of Higher Education I.M.Sechenov First Moscow State Medical University of the Ministry of Health of the Russian Federation
Acta Gastroenterol Latinoam 2017;47(3):182-189
Recibido: 21/09/2016 / Aprobado: 23/11/2016 / Publicado en www.actagastro.org el 02/10/2017
Summary
Some gastrointestinal disease are relationed with H pylori (Hp), a microorganism that is part of the gastric microbiota. It affects 50 to 90% of the world population. Objetive. Analyzed changes in the frequency of occurrence of LPS/O, VAC A antigens and high molecular weight proteins (HMWP, including CAG) of Hp and their specific immune complexes with IgG-class antibodies of inmunoglobulins (IgG-CIC) in patients with gastric diseases and volunteers in recent years. Material and methods. In the period of 2009-2016 samples of stools and circulating immune complexes (CIC) of serum samples of stools and circulating immune complexes (CIC) in 8568 patients with gastric diseases were evaluated and samples of stool, saliva and CIC of 23 volunteers were monitored and 14601 samples of biomaterials were studied. The reaction of coaglutination on glass and plates with specially prepared antibody diagnosis were used. LPS/O antigens of Hp werw detected in heated stool and saliva samples in reaction on glass; VAC A antigens and HMWP antigens in CIC composition of blood serum and saliva. Results. For the first time since year 2000, 27-55% increase (enhanced since 2009) the frequency of LPS/O antigens occurrence in the stool. It is a marker of Hp reproduction in the gastric mucosa. Additionally, more pronounced cyclical changes in the frequency of VAC A, CAG A detection, as well as a decrease in the frequency of virulence antigens was observed in the composition of CIC of blood serum and saliva, showing a 35-68% decrease in the body immune response. Conclusions. There is a increase in Hp infection due to decrease immunity to the Hp pathogenic factors, especially pronounced in 2012- the first half of 2016, indicating the possibility of increased risk of new diseases, exacerbations and adverse outcomes of Hp infection (peptic ulcer disease, atrophic gastritis, gastric cancer and gastric malt lymphoma).
Key words. Helicobacter pylori, LPS/O antigens, VAC A, CAG A, immunity, circulating inmune complexes.
Aumento de la infección por helicobacter pylori y disminución de la inmunidad en los últimos años
Resumen
Algunas enfermedades gastrointestinales (EGI) se relacionan a la infección por Helicobacter pilory (Hp), microorganismo que es parte de la microbiota del estómago. Infecta al 50- 90% de la población mundial con diferentes tasas de prevalencia según se trata de países desarrollados o en vías de desarrollo. Objetivo. Analizar los cambios en la frecuencia de aparición de antígenos LPS/O; VAC A y proteínas de alto peso molecular (HMWP, incluyendo CAG A) de Hp y sus complejos inmunes específicos con anticuerpos IgG-CIC en pacientes con EGI y voluntarios en los últimos años. Material y métodos. En el período 2009-2016 se realizaron pruebas en heces y de inmunocomplejos circulantes (CIC) en suero de 8.568 pacientes ambulatorios con EGI y se monitorearon muestras de heces, saliva y CIC de 23 voluntarios. Se estudiaron en total de 14.601 muestras de biomateriales. Se utilizó la reacción de coaglutinación sobre vidrio y placas con anticuerpos especialmente preparados. Se detectaron antígenos LPS/O de Hp en muestras de heces y saliva calentadas en reacción de vidrio, antígenos VAC A y HMWP en la composición CIC del suero y saliva. Resultados. Por primera vez desde el año 2000, se observó un aumento del 27-55% de antígenos LPS/O en las heces en la composición del CIC de suero y saliva, mostrando una relación de 35-68% de disminución de la respuesta inmune del cuerpo. Conclusiones. Existe un aumento de la infección por Hp debido a la disminución de la inmunidad a los factores patógenos de Hp, especialmente pronunciada en 2012 y la primera mitad de 2016, lo que indica la posibilidad de un mayor riesgo de nuevas enfermedades, exacerbaciones y resultados adversos de la infección por Hp (úlcera péptica, gastritis atrófica, cáncer y linfoma malt gástrico.
Palabras claves. Helicobacter pylori, LPS/O, antígenos VAC A, CAG A, inmunidad, inmunocomplejos circulantes.
Abbreviates
AII: acute intestinal infections
CagA: a marker of pathogenicity islet, co-oncogene
CIC: circulating immune complexes
CG: chronic gastritis
CGD: chronic gastroduodenitis
GID: gastrointestinal diseases
GU: gastric ulcer and duodenal ulcer
HMWP: high molecular weight proteins of H.pylori
IBS: irritable bowel syndrome
IgG-CIC: circulating immune complexes with IgG-class antibodies of immunoglobulin’s
Hp: Helicobacter pylori
LPS: lipopolysaccharide
VacA: vacuolating toxin of H.pylori
In the last decade, gastrointestinal tract diseases occupied a leading position in the list of human diseases. Majority of urban dwellers suffer from them in varying degrees, which is associated with the modern pace of life, stresses, unfavorable environmental situation and improper diet. Many gastrointestinal diseases (GID) are due to Helicobacter pylori (Hp) – a microorganism that is part of the indigenous stomach biota. It infects 50 to 90% of the Earth population, for the term of life persistence in the body and is in difficult relationship with it. This microbe has an extremely high variability, phase and genetic variations, which gives him ample capacity of adaptation, and the unique opportunity to evade the host defense mechanisms.1-3
The most studied virulence factors of Hp are vacuolating toxin VacA and cytotoxin-associated protein CagA. VacA is involved in formation of vacuoles in epithelial cells and their destruction, stimulates cytokine synthesis, and is a marker of inflammation. In penetration into the cell through a type IV secretion system, CagA, a marker of pathogenicity islet, causes structural changes in the actin and cytoskeleton of the cell, enhances apoptosis, inhibits cell-mediated immunity and is a co-oncogene.1, 4
Elective colonization of the stomach epithelium with Hp causes the development of chronic mucous membrane inflammation, which leads to the development of such common diseases as chronic gastritis, gastric ulcer and duodenal ulcer. Long-term persistence of Hp on gastric mucosa contributes to atrophic changes, apoptosis, intestinal metaplasia and the development of tumors. In 1994, WHO experts attributed Hp to the 1st risk group carcinogen developing stomach cancer. According to Eurogast Study Group, in the study of 3,194 asymptomatic individuals in 17 populations of Europe, USA, Japan, 10% increase of Hp-infection among the population leads to 27% increase in the prevalence of gastric cancer and 18% increase in mortality.5 Possible association of Hp with other non-gastric diseases is constantly being researched: vascular diseases, primary Raynaud’s syndrome, autoimmune diseases, Parkinson’s disease, certain skin diseases, and others.
Until now, despite intensive research, the key issues of helicobacter is remain poorly understood: Hp persistence mechanism and transition of asymptomatic chronic inflammation to periodic exacerbations, metaplasia and cancer. Of course, a great role in this process is played by factors of microbial pathogenicity, immune response of the body and external factors. Their effect can be evaluated only in massive and monitoring studies. However, multiple repeated studies over a long period of time using the existing complex, expensive, mainly invasive techniques is difficult, and sometimes impossible, for example, in children.
To perform multiple repeated studies of Hp pathogenicity factors in population in order to monitor Hp-infection and specific immunity to the pathogen, we have developed and tested non-invasive and cost-effective test systems for coagglutination reaction and methods for simultaneous determination of Hp-infection, strain virulence and evaluation of immunity. Long-term studies of patients with gastrointestinal diseases (GID) were conducted using them, and monitoring of asymptomatic (healthy) volunteers. Cyclical oscillation of frequency of Hp pathogenicity factor detection in the body was found as a result of these studies, different frequency (daily, appr. monthly, appr. yearly, long-term) and the pattern of changes in the ratio of identified Hp factors (LPS/O-antigen, VacA-antigen, cagA antigen),6-9 their dependence on certain internal and external factors (age, season of the year, month, year of research, type of disease),10 and the correlation of Hp antigen circulation with solar activity was shown for the first time (in its assessment by Wolf numbers); some factors and time periods of the risk of Helicobacter pylori infection exacerbations were identified.10, 11
The purpose of this study was to analyze the changes in the incidence of LPS/O-, VacA-, HMWP (including CagA) – Hp antigens and their specific immune complexes with IgG-class immunoglobulin antibodies (IgG-CIC) in patients with gastrointestinal diseases and asymptomatic volunteers in recent years.
Materials and methods
From 2009 to May (inclusive) 2016, we once examined 8,568 outpatients with a variety of gastrointestinal diseases (GID): chronic gastritis and gastroduodenitis (CG, CGD), gastric ulcer and duodenal ulcer (GU), irritable bowel syndrome (IBS), acute intestinal infections (AII) (5,614 stool samples and 2,948 serum samples), as well as 23 volunteers in weekly monitoring mode, including persons with a history of chronic gastroduodenitis and peptic ulcer disease (2,409 stool samples and 3,630 saliva samples). 14,601 samples of biomaterials were studied in total (the stool, saliva, blood serum).
The stool or saliva samples after preliminary heating (100 °C, 30 min) in the qualitative agglutination reaction on the glass, the presence of free LPS/O-antigen of Hp as a measure of Hp infection was determined. Antigens of high molecular weight proteins (HMWP, including CagA) and VacA were determined in the native samples of average molecular circulating immune complexes (IgG-CIC) in the semi quantitative on the plates. Production of test systems for determination of Hp antigens in QAR on the glass and plates in the N.F. Gamaleya Federal Research Center for Epidemiology and Microbiology of the Ministry of Health of the Russian Federation, preparation of bioassay for studying, and direct QAR on the glass and plates were described previously.12-15 Specially prepared natural highly specific hyper immune rabbit antiserum to O-antigen, HMWP complex (including CagA), as well as recombinant proteins of VacA Hp toxin were used for production of test systems.
Statistical data processing was carried out using parametric (Student’s t-test) and nonparametric (χ-square) analysis methods and correlation analysis in Statistica, Microsoft Word for Windows, Excel 2007, BioStat 2012 software. Differences at p < 0.05 were considered statistically significant.
Results
As a result of our research in 2009-2016, basic laws of cyclic change in the detection rate (occurrence) of O-, VacA- and HMWP (CagA)-Hp antigens in the body, established by us previously, have been confirmed: 1) daily fluctuations – with the low occurrence of free O-antigen at night and high occurrence during the day, while detection rate of VacA-antigens linked in IgG-CIC had the opposite trend; 2) seasonal fluctuations – with the highest detection rate of O-antigen in February-March and September-October (in all Hp-infected volunteers with small individual fluctuations); 3) fluctuations in O-antigen titers in the stool of patients with stomach diseases (non-ulcer dyspepsia, chronic gastritis, peptic ulcer disease, atrophic gastritis) higher than in asymptomatic volunteers, but with maintenance of the same dynamics; 4) opposite dynamics of detection rate of VacA-antigen and HMWP (CagA)-antigen in CIC as compared to detection of O-antigen in the stool, and each increase in O-antigen in the stool was accompanied by a decrease in VacA-antigen detection rate in CIC, and vice versa.
High levels of O-antigen in the stool clearly indicate increased vital activity of Hp and its reproduction, and detection of VacA-antigen, HMWP (CagA)-antigen in CIC indicates formation of specific antitoxic immunity.
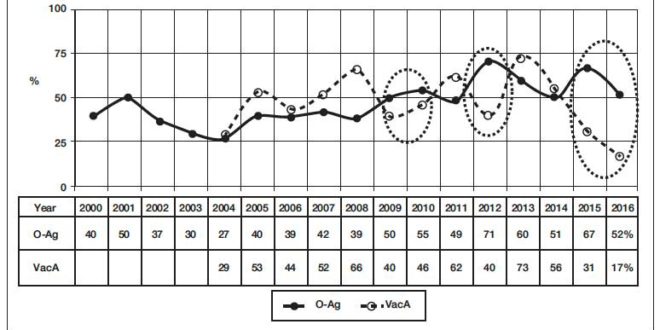
For the first time in research in 2009-2015, in contrast to our data of the earlier studies, since 2001, it was found that the average annual incidence of Hp O-antigen in the stool of GID patients significantly increased. If the incidence of O-antigen occurrence was 40% in 2000, in 2015 it was 67% (a 27% increase, p ≤ 0.01) (Figure 1). In chronic gastroduodenitis, in particular, the average detection rate of O-antigen has increased from 39% in 2000 to 89% in 2015 (50% increase, p ≤ 0.01) (Figure 2), and in patients with peptic ulcer and duodenal ulcer – from 35% to 90% (55% increase, p ≤ 0.01) (Figure 3).
Figure 1. Detection rate of O-antigen in the stool and VacA-antigen of Hp in CIC of blood serum in the screening of gastrointestinal diseases. (2000 – May 2016: 9,342 stool samples and 5,502 blood samples). (2009 – May 2016: 5,614 stool samples and 2,948 blood samples).
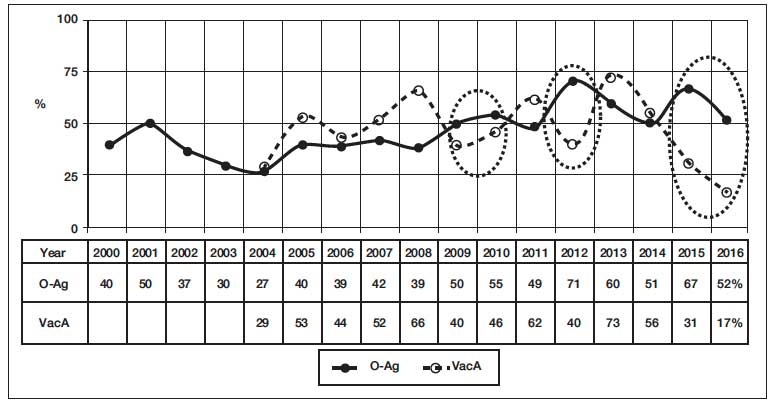
Figure 2. Detection rate of O and VacA-antigen of Hp in chronic gastroduodenitis (2000 – May 2016, 1,810 saliva samples).
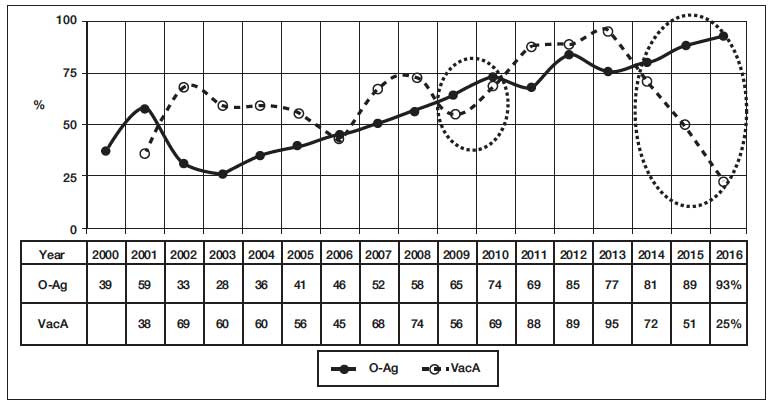
Figure 3. Detection rate of O and VacA-antigen of Hp in duodenal ulcer. (2000 – May 2016, 1,820 saliva samples).
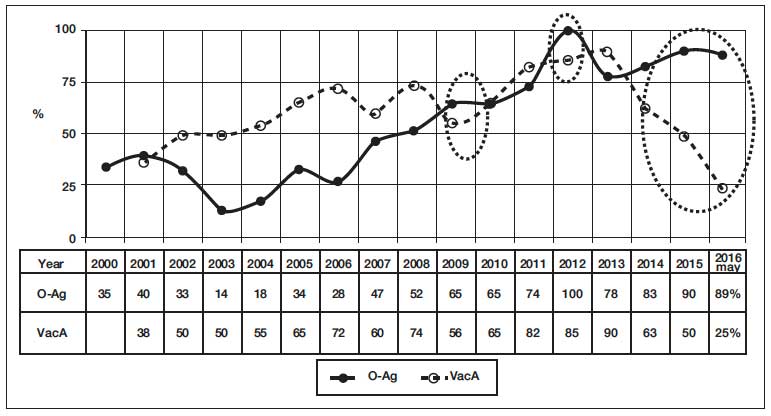
Gradual slow increase in the average detection rate of Hp O-antigen in the stool of in GID patients could be already observed since 2005, more significant – since 2009, and in some groups of patients, these dates were shifted slightly (Figure 1 – 3).
With increased infection and reproduction of Hp, determined by an increase in the detection rate of Hp O-antigen in the stool, in recent years, we have noted violent fluctuations in the detection rate of VacA-antigen in IgG-CIC, which in some years was higher than the detection rate of O-antigen, or was below it. And the latter evidenced of decreased immunity to this important Hp pathogenicity factor and to the microorganism in general. Such a reduction in immunity was observed in patients with GID in 2009-2010, 2012, 2015-2016.
The range of exceeding O-antigen detection rate in the stool over the detection rate of IgG in VacA-CIC in blood serum and saliva increased in recent years: in 2009, it amounted to an average of 9-10%, in 2012 – 15-31%, in 2015-2016 – 35-68%. This ratio showed a significant increase in the risk of new stomach diseases, exacerbations and possible severe complications with increased Hp vital activity in the intestine and lowered immunity to it.
The data obtained in 2009-2016 on the detection rate of VacA-antigen in IgG-CIC of blood serum or saliva differed sharply from that of the period from 2002 to 2007-2008, when the detection rate of VacA-antigen in IgG-CIC of blood serum or saliva constantly exceeded the detection rate of free O-antigen in the stool and saliva (Figure 1 – 3). In 2011 and 2013, there was a similar ratio of VacA-antigen in IgG-CIC and O-antigen values, indicating a sufficient immunity to Hp.
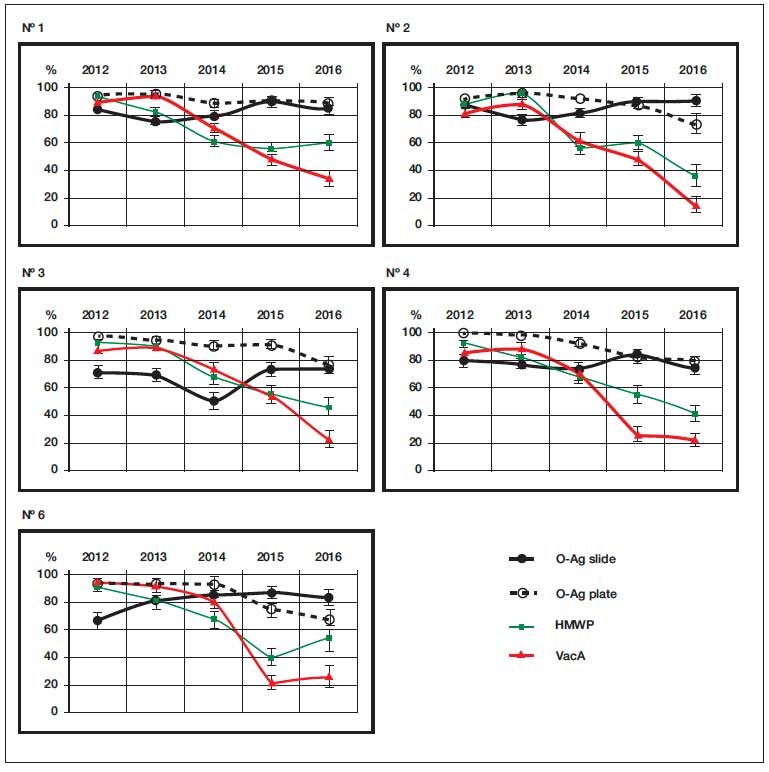
In asymptomatic Hp-infected volunteers, the survey data in long-term monitoring also showed a decrease in the detection rate of VacA-antigen in IgG-CIC in recent years against the constant or increased detection rate of O-antigen in the stool. Monitoring results of individual volunteers are shown in (Figure 4). As you can see, the detection rate of VacA-antigen in IgG-CIC, as well as the detection rate of HMWP (including CagA) was behind the detection rate of O-antigen in the stool in asymptomatic volunteers in recent years, indicating a decrease in Hp immunity, especially to the most important pathogenicity factors – VacA and CagA.
Figure 4. Monitoring of O-, VacA- and HMWP-antigens in Hp -infected volunteers (2000-2015, 1,662 saliva samples).
Discussion
The average annual detection rate of VacA in IgG-CIC of blood serum and saliva varies over a 15-years observation period and usually exceeds the annual average frequency of occurrence of O-antigen in the stool. In the years of reduction in VacA in IgG-CIC lower than O-antigen value in the stool, some patients had clinical exacerbation of diseases of the stomach and intestines – in 2001, 2009-2010, 2012, 2014-2016. The revealed regularities allow predicting the risks of exacerbations of inflammatory and erosive diseases of the stomach and duodenum.11
In recent years, instead of the expected decline in the rate of occurrence of O-antigen in the stool on the background of a sufficiently high content of VacA in IgG-CIC in 2013, the majority of surveyed persons had a significant decline in the level of formation of specific antiVacA- and antiHMWP- immune complexes in blood serum, which began in 2012, continued in 2015 and is passing to the first half of 2016. The decrease in the detection rate of VacA and CagA Hp virulence factors in IgG-CIC shows a significant reduction of antitoxic immunity in Helicobacter pylori infection, especially in recent years. It should be emphasized that these significant changes in anti-Helicobacter immunity occur against a background of the above growth in the detection rate of Hp O-antigen in the stool.
In view of the data published earlier on the increased risk of gastric cancer and death rates during the increase of Hp-infection of population,5 our data on the increase of Hp infection, especially in 2014-2016, and a sharp decrease in immunity to it in the form of a decrease in binding virulence antigens in circulating IgG-immune complexes indicate a severe, critical situation with Helicobacter pylori infection. In comparison with the beginning of observation period (2000), now there is a significant increase of Hp-infection in population (by 27-55%), phase changes in Hp pathogenicity, and decreased antitoxic immunity (by 35-68%), which poses serious health risks to the health of population because of the greater probability of the emergence of new stomach diseases in Hp-infected individuals, greater severity of exacerbations and complications in acute and chronic gastrointestinal tract diseases, and acceleration of carcinogenesis processes in the future.
Thus, 15-years screening tests of Hp persistence in the body of patients with gastrointestinal diseases were conducted for the first time, as well as monitoring of asymptomatic volunteers using non-invasive immunoassay (coagglutination reaction) with detection of the incidence of O-, VacA- and CagA-antigens of Hp in various biosubstrates (the stool, saliva, blood serum) in free form and in IgG-CIC. The result revealed cyclical changes in these indicators, on the one hand reflecting vital activity of the causative agent (infection, reproduction, production of toxins or other pathogenicity factors), and on the other hand, the state of the host immune system, their mutual adaptation to internal and external factors (the dominant among them are obviously the immunity and solar activity).
Periodic changes of vital activity (reproduction) and microbial pathogenicity, as well as immune responses in chronic Hp persistence, while taking solar activity and other factors into account, should be considered in medical practice in diagnostics, treatment, prognosis and prevention of Hp infection, in individual management of patients in personalized medicine.
Given the unconditional role of Hp in pathogenesis of various gastrointestinal diseases, including gastritis, peptic ulcer disease and gastric cancer, and the alarming situation relating to increased Hp infection and decreased immunity of population to this pathogen in recent years, it is necessary to continue such long-term informative non-invasive research to study pathogenesis of Hp infection and to develop urgent measures on prevention, treatment and monitoring of treatment effectiveness, as well as to obtain new scientific data on the mechanisms of persistence of Hp and other microbes.
Conflicts of interest. The authors declare no conflict of interest.
References
- Salama NR, Hartung ML, Müller A. Life in the human stomach: persistence strategies of the bacterial pathogen Helicobacter pylori. Nat Rev Microbiol 2013; 11: 385-399.
- Zhebrun AB. Infection with Helicobacter pylori. S-Pb, «Phoenix» 2006. 380 p.
- Lina TT, Alzahrani S, González J, Pinchuk IV, Beswick EJ, Reyes VE. Immune evasion strategies used by Helicobacter pylori. World J Gastroenterol 2014; 20: 12753-12766.
- Brawner KM, Morrow CD, Smith PD. Gastric Microbiome and Gastric Cancer. Cancer J 2014; 20: 211-216.
- The EUROGAST Study Group. Epidemiology of, and risk factors for, Helicobacter pylori infection among 3194 asymptomatic subjects in 17 populations. Gut 1993; 34: 1672-1676
- Belaya Yu A, Vakhrameeva MS, Petrukhin VG, Bondarenko VM, Belaya OF, Evdokimov VV, Kurmanova DM, Iudina TI, Nesterenko VG. Occurrence of Helicobacter pylori specific antigens in diseases of gastrointestinal tract. Zh Mikrobiol Epidemiol Immunobiol 2004; 6: 63-69.
- Belaya Yu A, Belaya OF, Petrukhin VG, Bystrova SM, Vakhrameeva M S, Cyclical fluctuations in the frequency of occurrence of Helicobacter pylori antigens in the long persistence in the body. «The collective monograph: The results of the science». Selected Works of the International symposium on fundamental and applied problems of science. Moscow: Russian Academy of Sciences 2013; V.3, Chapter 9: 146-157.
- Belaya Yu A, Belaya OF, Petrukhin VG, Vakhrameeva MS, Bystrova SM, Pronin AV. Immunologic monitoring of Helicobacter pylori persistence in the organism. Zh Mikrobiol Epidemiol Immunobiol. Moscow 2015; V. 4: 106-112.
- Belaya Yu A, Petrukhin VG, Vakhrameeva MS, Belaya Yu F, Belaya OF. A method for determining probability of Helicobacter pylori infection. Patent RUS Nº 2360251.
- Belaya Yu A, Belaya OF. Detection rate of Helicobacter pylori O- and VacA-antigens in children and adults with gastrointestinal diseases in comparison with the solar activity. X International Crimean Conference “COSMOS AND BIOSPHERE” 2013;182-183.
- Belaya Yu A, Petrukhin VG, Belaya OF. The method for determining the risk of exacerbations of Helicobacter pylori infection. Patent RUS 2481584.
- Belaya OF, Cherkasov VL, Belaya Yu A, Bystrova SM, Tseneva GY, Vyalbe EV. Coagglutination reaction at infectious diseases. Guidelines. Moscow 1990: 1-12.
- Belaya Yu A, Belaya OF, Petrukhin VG. A method for producing diagnostic kit for the detection of Helicobacter pylori antigens in coagglutination reaction. Patent RUS 2186394.
- Belaya Yu F, Sheklakova LA, Vakhrameeva MS, Zhukhovitskii VG, Petrukhin VG, Belaya Yu A. Production of recombinant fragment of VacA protein and development of non-invasive method for the diagnosis of Helicobacter pylori infection. Mol Gen MikrobiolVirusol. Moscow 2005; 1: 32-35.
- Belaya Yu A, Vakhrameeva MS, Belaya Yu F, Belaya OF, Petrukhin VG. A method for producing a test system for the determination of protein antigens of citotoxin-associated proteins of Helicobacter pylori in biological material of infected individuals in coagglutination reaction. Patent RUS Nº2232989.
Correspondencia: Olga F. Belaya
2-4 Bolshaya Pirogovskaya st., Moscow 119991, Russia
Correo electrónico: ofbelaya@mail.ru
Acta Gastroenterol Latinoam 2017;47(3): 182-189
 Revista ACTA Órgano Oficial de SAGE
Revista ACTA Órgano Oficial de SAGE