Wandregíselo Ponce de Leon Júnior1 ID · Riccardo Pratesi2 ID· Felipe Mendes Dos Santos Cardia3 ID· Lenora Gandolfi4 ID· Rosa Harumi Uenish5 ID
1 Programa de Pós-Graduação em Ciências da Saúde da Universidade de Brasília.
2 Laboratório de Doença Celíaca, Universidade de Brasília – Médico pesquisador.
3 Universidade de Brasília – Profesor.
4 Programa de Pós-Graduação em Ciências da Saúde da Universidade de Brasília – Orientadora.
5 Laboratório de Doença Celíaca, Universidade de Brasília – Pesquisadora.
Brasília, Brasil.
Acta Gastroenterol Latinoam 2021;51(4):413-419
Received: 11/10/2021 / Accepted: 15/11/2021 / Published in www.actagastro.org 13/12/2021 / https://doi.org/10.52787/SUYI2286
Summary
Introduction. Celiac disease is a chronic immune-mediated enteropathy precipitated by the ingestion of gluten contained in wheat, barley and rye, which occurs in individuals with genetic susceptibility. These patients may have iron deficiency anemia as the only symptom. Aim. To determine the prevalence of newly diagnosed celiac disease in patients undergoing treatment for iron deficiency anemia. Material and methods. Cross-sectional study. Patients from the hematology outpatient clinic of a tertiary hospital in Brasília were included between October 2017 and March 2019. The patients were older than 18 years, with a diagnosis of iron deficiency anemia of unknown etiology. The sample consisted of 1,034 patients, and 184 met the inclusion criteria. Serum levels of IgA, IgA anti-transglutaminase, and IgA anti-endomysial antibodies were measured. For each positive screening, intestinal biopsies were performed. Results. Of the 184 patients, six (3.26%) presented celiac disease. These patients had a median age of 24 years, with an age range from 23 to 49 years, all females. Conclusion. The prevalence of celiac disease in undiagnosed patients with iron deficiency anemia in Brasília, Federal District is high (1:33).
Keywords. Celiac disease, anemia, iron deficiency, transglutaminases.
Prevalencia de enfermedad celíaca en pacientes con anemia por deficiencia de hierro en Brasilia, Brasil: un estudio de corte transversal
Resumen
Introducción. La enfermedad celíaca es una enteropatía crónica inmunológica precipitada por la ingestión del gluten contenido en el trigo, la cebada y el centeno, que ocurre en individuos con susceptibilidad genética. Estos pacientes pueden tener anemia por deficiencia de hierro como único síntoma. Objetivo. Determinar la prevalencia de enfermedad celíaca de reciente diagnóstico en los pacientes con anemia por deficiencia de hierro. Material y métodos. Estudio de corte transversal. Se incluyeron pacientes del servicio ambulatorio de hematología de un hospital terciario de Brasilia, entre octubre de 2017 y marzo de 2019. Los pacientes incluidos fueron mayores de 18 años, con diagnóstico de anemia por deficiencia de hierro de origen desconocido. La muestra consistió en 1.034 pacientes, de los cuales 184 cumplieron con los criterios de inclusión. Se determinaron los niveles de IgA en suero, IgA anti-transglutaminasa y anticuerpos anti-endomisio IgA. Para cada resultado positivo, se realizaron biopsias intestinales. Resultados. De los 184 pacientes, seis (3,26%) presentaron enfermedad celíaca. Estos pacientes tuvieron una mediana de edad de 24 años, con un rango de edad de 23 a 49 años, todas mujeres. Conclusión. La prevalencia de enfermedad celíaca entre pacientes diagnosticados con anemia por deficiencia de hierro en Brasilia, Distrito Federal, es alta (1:33).
Palabras claves. Enfermedad celíaca, anemia, deficiencia de hierro, transglutaminasas.
Abbreviations
CD: Celiac Disease.
HLA: Histocompatibility leukocyte antigen.
IDA: Iron deficiency anemia.
BMI: Body mass index.
MCV: Mean corpuscular volume.
IgA: Immunoglobulin A.
IgA-tTG: Tissue transglutaminases antibody.
IgA-EMA: Endomysial antibody.
ELISA: Enzyme-linked immunosorbent assay.
GI: Gastrointestinal.
Introduction
Celiac disease (CD) is a chronic immune-mediated enteropathy precipitated by the ingestion of the gluten contained in wheat, barley, and rye, in genetically susceptible individuals. Histocompatibility leukocyte antigens (HLA) DQ2 and DQ8 are the main genetic predisposing factors, and are found in at least 95% of patients with celiac disease. CD is currently a worldwide problem and one of the most common lifelong disorders, with an estimated seroprevalence of 1.4%, which varies from one continent to another. However, due to its low specificity, seroprevalence may overestimate the prevalence of CD. On the other hand, the prevalence of CD diagnosed by biopsy was estimated at 0.7%, also revealing continental and regional variations. This can be considered an underestimation of the condition, since not all individuals undergoing serological screening usually consent to a complete endoscopic evaluation.1 Current treatment is based on strict adherence to a gluten-free diet, which, although not successful in all patients, results in histological improvement and the remission of gluten-specific antibodies.2
Once a rare disorder, characterized primarily by malabsorption, CD has become more common and occurs at any age, from early childhood to older adults. Diagnosis of CD is quite difficult, as symptoms can vary significantly from patient to patient, affecting different age groups. Intestinal CD is detected more frequently in the pediatric population. The predominant symptoms are diarrhea, loss of appetite, abdominal distension and failure to thrive; other symptoms are vomiting, irritability, anorexia and, less frequently, constipation. Adult CD, on the other hand, is generally associated with symptoms suggestive of irritable bowel syndrome and dyspepsia, followed by nausea and, to a lesser extent, vomiting.3
Extraintestinal symptoms are common in children and adults. Short stature, fatigue and headache are the most common extraintestinal symptoms in children. In adults iron deficiency anemia (IDA), fatigue, headaches, and psychiatric disorders are common extraintestinal manifestations. Other manifestations detected only in adults, are alopecia, infertility, peripheral neuropathy, and osteoporosis.3, 4
IDA is the most common form of microcytic anemia. It is assumed that 50% of the cases are due to iron deficiency.5 Occult intestinal bleeding, hypermenorrhea and reduced iron absorption are the most common causes of IDA.6 Anemia is one of the most frequent extraintestinal manifestations of CD, often the main clinical feature of the disease.7, 8 IDA has been reported in up to 46% of cases of non-classic CD, with a higher prevalence in adults compared to children.9 The reported prevalence of CD in patients with IDA varies significantly worldwide. An extensive meta-analysis, spanning several countries,10 found that biopsy confirmed CD in 1 out of 31 patients with IDA, on average, justifying the consensus that testing should be performed in all IDA patients.11
IDA usually involves both increased iron loss and decreased iron absorption.7 Iron absorption occurs in the small intestine and depends on several factors, such as the integrity of the epithelial mucosa and intestinal acidity. Iron deficiency anemia usually is a microcytic and hypochromic anemia. Patients characteristically have low serum iron levels, high total binding capacity, and low ferritin levels.12, 7 Iron deficiency in CD is primarily the result of impaired absorption of iron due to alterations of the mucosal epithelium. Occult gastrointestinal bleeding is another cause and has been detected in patients with CD, depending on the degree of villous atrophy.13, 14
Undiagnosed CD can result in serious consequences and significantly high morbidity15 and mortality.16 The duration of CD diagnosis is still unacceptably long17 so many cases remain undetected without active serologic screening18. Generally, CD patients have long-standing symptoms and consult many physicians before diagnosis 19. In a major study by Green et al.20, which included patients with biopsy-proved CD, the median delay to CD diagnosis was 11 years. Although IDA is often the initial complaint of a nonclassical/extraintestinal CD21, most physicians rarely include serologic testing for CD as part of their initial studies.22
Despite the aforementioned facts, the frequency of CD in the Brazilian population with IDA was never investigated, which justifies the purpose of this study. The aim was to determine the prevalence of CD, among undiagnosed patients, undergoing treatment for IDA in the hematology clinic of a tertiary hospital.
Methods
This was a cross-sectional observational study conducted in IDA patients. The sample size (n = 184) was calculated using Epi Info version 3.5.2, considering a confidence interval of 95%, a margin of error of 5.1 and a predicted frequency of 14.6%.23
The research was carried out with consecutive IDA patients, who attended the hematology outpatient clinic of a tertiary hospital in Brasília. This hospital is part of the tertiary hospital network that predominantly serves the low-income population mostly dependent on the Brazilian Unified Health System.
The inclusion criteria were individuals with IDA and aged over 16 years. The hemoglobin level adopted for anemia was <13 g/dL in men and <12 g/dL in women, MCV < 80 fl (femtoliters) and ferritin levels < 30 ng/mL.24 No previous examination was performed among eligible subjects for the presence of symptoms or conditions, commonly associated with CD.
Subjects who presented any of the following criteria were excluded: men with actively bleeding or recent history of melena, hematemesis, hematochezia, hemoptysis, recurrent epistaxis, hematuria, trauma, previous diagnosis of CD, history of cancer, chronic kidney, liver or heart diseases; and women with hypermenorrhea with a cycle longer than 7 days, history of metrorrhagia, previous gastrointestinal surgery and an ongoing pregnancy.
According to the Declaration of Helsinki, all patients received written and verbal information regarding the objective of the study and written informed consent was obtained from all participants. The study protocol was approved by the Ethics Committee of the Health Sciences Teaching and Research Foundation (CAAE: 75375817.7.0000.5553).
The diagnosis of CD must meet the following criteria positive IgA, IgA-tTG, and IgA-EMA test suggest an intestinal biopsy. IgA tests were performed in a private laboratory using the turbidimetric immunoassay (COBAS MIRA™, Roche Diagnostic Systems). IgA-tTG test, used as the first-level screening step, was determined by enzyme-linked immunosorbent assay (ELISA) using a commercial kit (QUANTA Lite™ h-tTG IgA ELISA, INOVA Diagnostic Inc., San Diego, CA, U.S.A.). All positive finding were confirmed by the IgA-EMA assay by indirect immunofluorescence (QUANTA LITE™ Endomysial Primate Distal Esophageous Slide kit, INOVA Diagnostic, Inc. San Diego, CA, USA) and two independent observers examined each slide. A flowchart displaying various stages of the study is represented in Figure 1.
Figure 1. Flowchart showing the various phases that each patient with IDA underwent during the study.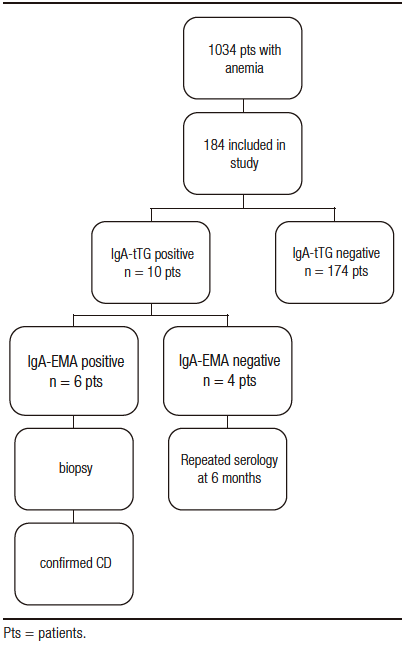
All positive serologic test results (IgA-tTG and IgA-EMA) underwent upper endoscopy to ensure higher sensitivity and specificity for the diagnosis of CD, due to strong evidence that both IgA-tTG and IgA-EMA are associated with high sensitivity and specificity, above 95%.25 Duodenal biopsies were performed using an AOHUA® AQ-100 Endoscopic System appliance; five samples were taken under direct visualization, from the second portion of the duodenum. The biopsy samples were evaluated by a pathologist with extensive experience in gastrointestinal disorders and graded according to Marsh-Oberhuber classification.26
Patients with IGA-tTG levels between 20 and <90 were instructed to continue the gluten diet and were reevaluated six months and one year later. After this period, IgA-EMA and IgA-tTG tests were repeated. If positive, biopsies were taken.
The Mann-Whitney test was performed and the p value of <0.05 was adopted for a significant association of hemoglobin, body mass index (BMI), and transglutaminase variables.
Results
A total of 1,034 patients were interviewed between October 2017 and March 2019.
Among the 1034 patients (see Figure 1), 184 consenting participants after applying the exclusion criteria were included. Overall, they were 21 males and 163 females with a median age of 45 years (18-80). The ratio between female and male was 7.8.
Serologic Tests and Biopsy Results
All serum IgA levels were within normal limits. Ten patients had a positive IgA-tTG. Four of them had IgA-tTG values >20 and <90 and were negative for IgA-EMA. These patients were advised to continue their normal diet and were scheduled for new serologic retesting within six months. They had a follow-up consultation every six months for one year. All had a negative IgA-EMA result and biopsies classified as Marsh 0. After this period, the diagnosis of CD was discarded.
Six patients (3.26%) were positive for both IgA-tTG (values > 100) and IgA-EMA. The results of the serologic tests are illustrated in Table 2. All six subjects positive for IgA-tTG and IgA-EMA underwent upper GI endoscopy. Biopsy results were graded according to the Marsh-Oberhuberand classification and can be seen in Table 2.
The most frequent symptoms reported by the six CD positive patients were headache (66.7%), abdominal pain (50%), and other gastrointestinal symptoms (flatulence, postprandial fullness, abdominal pain and, diarrhea) (33,3%). The median age of the patients diagnosed with CD was 24 years (range from 23 to 49 years), all of them female (100%). The mean duration of CD diagnosis among these 6 patients from the onset of anemic symptoms to gastrointestinal symptoms was 5 years. Hemoglobin and BMI of patients with and without CD revealed no significant differences (Table 1).
Table 1. Descriptive measurements of hemoglobin and BMI values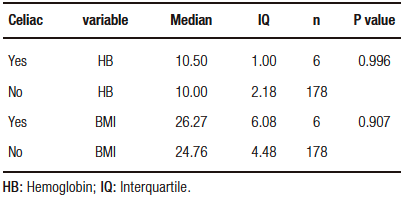
Table 2. Data from the celiac patients with laboratory and serologic results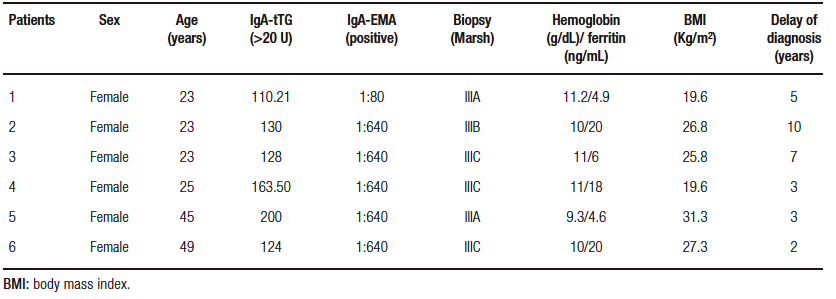
Discussion
The study was conducted in a hematology outpatient clinic of a tertiary hospital in Brasília, Federal District, Brazil and is geographically representative of people all over Brazil. In terms of race, it is a racially diverse study population with a considerable number of patients from European descent, with a variable inclusion of other races, mainly black and to a lesser extent of Amerindians.
IDA is the most common form of anemia in the world and may or may not be associated with other diseases. CD is one of the diseases with a high prevalence of IDA, as reported in studies by Sansotta et al.27 and De Falco et al.28 with a prevalence of 48% and 45% respectively.
On the other hand, studies on the prevalence of CD in patients with IDA report a higher prevalence of CD in these patients than in the general population. This is confirmed in a systematic review and meta-analysis performed by Mahadev10 that identified eighteen studies with 2,998 patients (adults and children) reporting a prevalence of 3.1 to 5.5% of CD in patients with IDA. This finding is almost similar to that observed in our study on the prevalence of CD among patients with IDA, reported as 1:33 (3.26%).
Despite being like our study, the prevalence of CD in Brazil (Brasília) was 3,41 per 1,000 individuals.29 However, it was not performed in patients with anemia but in the general population.
Other studies on the prevalence of CD in anemic patients have alternate results, with values ranging between 0 and 10.4%, as shown in studies performed in Iran and Turkey.30, 31 These differences with our study are probably explained by the regional peculiarities of each country and disparities in the diagnostic criteria used. Some studies consider biopsy to be the the gold standard method for diagnosing CD, even though they include IgA-tTG analysis but without IgA-EMA testing.
In this study, 2.17% of patients with IgA-tTG had positive values (>20 and <90). These patients were instructed to maintain their gluten diet and were re-evaluated at six months and one year. All had a negative IgA-EMA result and intestinal biopsies classified as Marsh 0. After this period, the diagnosis of CD was discarded. This drew our attention to the false positive of IgA-tTG antibodies, as stated by Caio.3 Diseases such as liver disease, rheumatoid arthritis, giardiasis, and inflammatory bowel disease can lead to false positives in IgA-tTG testing.30-32 Studies highlight that the sensitivity and specificity of IgA-tTG is considered one of the most sensitive serologies for the diagnosis of CD, while IgA-EMA is one of the most specific, therefore, it is used for diagnostic confirmation.33 A low antibody titer for IgA-tTG, not associated with duodenal mucosal damage, has been reported in some cases of autoimmune diseases, infections, tumors, myocardial lesions, psoriasis, some liver diseases, and rheumatoid arthritis.30, 34
Khatoon, Ahmed, and Yousaf35 described a 75% prevalence of CD in individuals under 40 years of age, with a predominance of CD in individuals between 20 and 30 years old, with a percentage of 11.81%. The sociodemographic data (age and gender) of our study do not differ from those presented in the current literature, with a predominance of CD in individuals between 21 to 30 years old, with a percentage of 66.7%.
CD is more common in women, with a ratio of 2:1 or 3:1 compared with men, which is not different from other autoimmune diseases.36 In this study, we observed a total female predominance (100%) of CD. This may be explained by the fact that the sample was composed of 88.6% of women.
Studies by Ganji et al.37 and Baghbanian et al.30 did not delineate a statistically significant correlation between hemoglobin and ferritin levels in patients with CD and their histopathological changes. This study is like those of Ganji et al.37 and Baghbanian et al.30 since it was not possible to establish a relationship between the severity of anemia and intestinal mucosa injury, according to the Marsh classification. The study by Berry et al.38 confirmed the hypothetical correlation between the severity of anemia and the degree of intestinal atrophy, although it was not proven in this study probably due to some methodological differences and characteristics of the population studied.
Zipser et al.39 conducted a nationwide survey involving 1,032 respondents with a mean age of 46 years with CD at diagnosis. Adult participants reported some gastrointestinal symptoms and 21% reported symptoms for more than 10 years prior to diagnosis.39 Another study conducted in Germany40 reported that the interval from the onset of symptoms to the first visit to the physician was greatly surpassed by the interval between the first visit to the physician and time of diagnosis. Other authors also observed a long duration of symptoms prior to definitive diagnosis of CD.19, 20, 40, 41 In this study, the patients reported bloating, headache, abdominal pain, and postprandial fullness, however, the main symptom prior to a definitive diagnosis was anemia. Paez et al.21 studied the duration of CD diagnostic among 101 adults, 52 with gastrointestinal symptoms and 49 symptomless. Anemia was reported in 11.5% of those presenting gastrointestinal symptoms. The duration of diagnostic among subjects presenting with a gastrointestinal symptom was approximately 2.3 months.
The time between the onset of gastrointestinal symptoms and definitive diagnosis of CD, in this study, was a mean of five years, which is relatively long by current standards. Many reasons could account for this delay between symptoms onset and diagnosis; however, it is logical to assume that the lack of awareness of non-gastroenterologist physicians about a potential CD is likely, especially in monosymptomatic cases. CD is always considered the responsibility of gastroenterologists.
There could be a selection bias in this study since the study participants were only selected in the hospital premises, precisely the hematology sector, considering that women have more frequent disturbances in iron absorption, either by loss of iron or associated diseases compared to men.
Clearly, this study had a small sample size, given that only patients with IDA who were recruited from a local hospital hematology clinic within a limited time period and who met the inclusion criteria were included. Thus, the results are not representative of the national territory. For all these reasons, multicenter studies are recommended for a more representative sample.
Conclusion
In conclusion, the prevalence of undiagnosed/uninvestigated CD in patients with IDA in Brasília, Federal District, is high (1:33). Despite the evolution of modern medicine, there is still a huge gap between the onset of CD symptoms and definitive diagnosis, with the risk of future complications to the patient’s health.
Key points
– IDA is the most common form of anemia in the world and may or may not be associated with other diseases.
– CD is one of the diseases with a high prevalence of IDA.
– We should always be suspicious of the possibility of CD in patients with IDA.
Consent for Publication. Anonymized data were used for the elaboration of this article, which did not distort its scientific value.
Intellectual Property. The authors declare that the data, figures and tables that appear in this article are original and were made in their belonging institutions.
© 2021 Acta Gastroenterológica latinoamericana. This is an open-access article released under the terms of the Creative Commons Attribution (CC BY-NC-SA 4.0) license, which allows non-commercial use, distribution, and reproduction, provided the original author and source are acknowledged.
Cite this article as: Ponce de Leon Júnior W, Pratesi R, Mendes Dos Santos Cardia F, et al. Prevalence of Celiac Disease in Patients with Iron Deficiency Anemia in Brasilia, Brazil: A Cross-Sectional Study. Acta Gastroenterol Latinoam. 2021;51(4):413-9. https://doi.org/10.52787/SUYI2286
References
- Lebwohl B, Rubio-Tapia A. Epidemiology, Presentation, and Diagnosis of Celiac Disease, Presentation. Gastroenterology 2021;160:63-75. 10.1053/j.gastro.2020.06.098.
- Cichewicz AB, Mearns ES, Taylor A, et al. Diagnosis and treatment patterns in celiac disease. Dig Dis Sci 2019;64:2095-2106. 10.1007/s10620-019-05528-3.
- Caio G, Volta U, Sapone A, Leffler DA, De Giorgio R, Catassi C, Fasano A. Celiac Disease: A comprehensive current review. BMC Med 2019;17:142. 10.1186/s12916-019-1380-z.
- Jericho H, Sansotta N, Guandalini S. Extraintestinal manifestations of celiac disease: Effectiveness of the gluten-free diet. J Pediatr Gastroenterol Nutr 2017;65:75-79 – 2017. Extraintestinal Manifestations of Celiac Disease: Effectiveness of the Gluten-Free Diet. J Pediatr Gastroenterol Nutr 65:75-79. 10.1097/MPG.0000000000001420.
- Lopez A, Cacoub P, Macdougall IC, Peyrin-Biroulet L. Iron deficiency anaemia. Lancet 2016; Feb 27, 387, S0140673615608650–:907-916. 10.1016/S0140-6736(15)60865-0.
- Talarico V, Giancotti, Mazza GA, Miniero R, Bertini M. Iron deficiency anemia in celiac disease. Nutrients 2021;13:1695. 10.3390/nu13051695.
- Halfdanarson TR, Litzow MR, Murray JA. Hematologic manifestations of celiac disease. Blood 2007;109: 412-421. 10.1182/blood-2006-07-031104: 10.1182/blood-2006-07-031104.
- Martín-Masot R, Nestares MT, Diaz-Castro J, López-Aliaga I, Alférez MJM, Moreno-Fernandez J, Maldonado J. Multifactorial etiology of anemia in celiac disease and effect of gluten-free diet: A comprehensive review. Nutrients 2019;11. 10.3390/nu11112557.
- Stefanelli G, Viscido A, Longo S, Magistroni M, Latella G. Persistent iron deficiency anemia in patients with celiac disease despite a gluten-free diet. Nutrients 2020;12:2176. 10.3390/nu12082176.
- Mahadev S, Laszkowska M, Sundström J, Björkholm M, Lebwohl B, Green PHR, Ludvigsson JF Prevalence of celiac disease in patients with iron deficiency anemia-A systematic review with meta-analysis. Gastroenterology 2018;155:374-382.e1. 10.1053/j.gastro.2018.04.016.
- Ludvigsson JF, Bai JC, Biagi F, et al. Diagnosis and Management of adult Coeliac Disease: Guidelines from the British Society of Gastroenterology. Gut 2014;63:1210-1228. 10.1136/gutjnl-2013-
306578. - Cook JD. Diagnosis and management of iron-deficiency anaemia. Best Pract Res Clin Haematol 2005;8: 319-332. 10.1016/j.beha.2004.08.022.
- Mant MJ, Bain VG, Maguire CG, Murland K, Yacyshyn BR. Prevalence of occult gastrointestinal bleeding in celiac disease. Clin Gastroenterol Hepatol 2006 4:451-454. 10.1016/j.cgh.2005.12.010.
- Shamir R, Levine A, Yalon-Hacohen M, et al. Faecal occult blood in children with coeliac disease Eur J Pediatr 2000;159:832-834. 10.1007/PL00008348.
- Choung RS, Larson SA, Khaleghi S, et al. Prevalence and Morbidity of Undiagnosed celiac disease from a Community based study. Gastroenterology 2017;152:830-839.e5. 10.1053/j.gastro.2016.11.043.
- Rubio-Tapia A, Kyle RA, Kaplan EL, et al. Increased prevalence and mortality in undiagnosed celiac disease. Gastroenterology 2009;137:88-93. 10.1053/j.gastro.2009.03.059.
- Norström F, Lindholm L, Sandström O, Nordyke K, Ivarsson A. Delay to celiac disease diagnosis and its implications for health-related quality of life. BMC Gastroenterol 2011;11:118. 10.1186/1471-230X-11-118.
- Lionetti E, Catassi C. Co-localization of gluten consumption and HLA-DQ2 and –DQ8 genotypes: A clue to the history of celiac disease. Dig Liver Dis 2014;46:1057-1063. 10.1016/j.dld.2014.08.002. Epub Sep 4.
- Dickey W, McConnell JB. How many hospital visits does it take before celiac sprue is diagnosed? J Clin Gastroenterol 1996;23:21-23. 10.1097/00004836-199607000-00007.
- Green PHR, Stavropoulos SN, Panagi SG, Goldstein SL, Mcmahon DJ, Absan H, Neugut AI. Characteristics of adults celiac disease in the USA: Results of a national survey. Am J Gastroenterol 2001;96: 126-131. 10.1111/j.1572-0241.2001.03462.x.
- Paez MA, Gramelspacher AM, Sinacore J, Winterfield L, Venu M. Delay in Diagnosis of celiac disease in patients without gastrointestinal Complaints. Am J Med 2017;130:1318-1323. 10.1016/j.amjmed.2017.05.027.
- Smukalla SM, Lebwohl B, Mears JG, Leslie LA, Green PH. How often do hematologists consider celiac disease in iron-deficiency anemia? Results of a national survey. Clin Adv Hematol Oncol 2014;12:100-105. PMID: 24892255.
- Zamani F, Mohamadnejad M, Shakeri R, et al. Gluten sensitive enteropathy in patients with iron deficiency anemia of unknown origin. World J Gastroenterol 2008;14:7381-7385. 10.3748/wjg.14.7381.
- OMS 2014. Organização mundial da saúde 2014. Protocolo Clínico e Diretrizes Terapêuticas, anemia por deficiência ferro. Portaria SAS/Ms 247.
- Al-Toma A, Volta U, Auricchio R, et al. European Society for the Study of Coeliac Disease (EScCD) guideline for coeliac disease and other gluten-related disorders. U Eur Gastroenterol J 2019;7:583-613. 10.1177/2050640619844125.
- Oberhuber G. Histophatology of celiac disease. Biomed Pharmacother 2000;54:368-372. 10.1016/S0753-3322(01)80003-2.
- Sansotta N, Amirikian K, Guandalini S, Jericho H. Celiac disease symptom resolution: Effectiveness of the gluten-free diet. J Pediatr Gastroenterol Nutr 2018;66:48-52. 10.1097/MPG.0000000000001634.
- De Falco L, Tortora R, Imperatore N, et al. The role of TMPRSS6 and HFE variants in iron deficiency anemia in celiac disease. Am J Hematol 2018;93:383-393. 10.1002/ajh.24991.
- Pratesi R, Gandolfi L, Garcia SG, Modelli IC, Lopes de Almeida P, Bocca AL, Catassi C. Prevalence of coeliac disease: Unexplained age-related variation in the same population. Scand J Gastroenterol 2003;38:747-750. 10.1080/00365520310003255.
- Baghbanian M, Farahat A, Vahedian HA, Sheyda E, Zare-Khormizi MR. The Prevalence of celiac disease in Patients with iron-deficiency anemia in center and South area of Iran. Arq gastroenterol 2015;52:278-282. 10.1590/S0004-28032015000400006.
- Grisolano SW, Oxentenko AS, Murray JA, Burgart LJ, Dierkhising RA, Alexander JA. The use fullness of routine small bowe biopsies in evaluation of iron deficiency anemia. J Clin Gastroenterol 2004;38:756-760. 10.1097/01.mcg.0000139034.38568.51.
- Emami MH, Karimi S, Kouhestani S. Is routine duodenal biopsy necessary for the detection of celiac disease in patients presenting with iron deficiency anemia? Int J Prev Med 2012;3:273-277. PMID: 22624084.
- Volta U, Granito A, Fiorini E, et al. Usefulness of antibodies to deamidated gliadin peptides in celiac disease diagnosis and follow-up. Dig Dis Sci 2008;53:1582-1588. 10.1007/s10620-007-0058-0.
- Husby S, Koletzko S, Korponay-Szabó I, et al. European Society Paediatric gastroenterology, Hepatology and Nutrition guidelines for diagnosing coeliac disease 2020. J Pediatr Gastroenterol Nutr 2020;70:141-156.
- Khatoon S, Ahmed A, Yousaf S. Iron Deficiency anaemia in Pakistan: Celiac disease an Underlying cause. J Ayub Med Coll Abbottabad 2018;30:372-376. PMID: 30465368.
- Ludvigsson JF, Card TR, Kaukinen, K, Bai J, Zingone F, Sanders DS, Murray JA. Screening for celiac disease in the general population and in high-risk groups. U Eur Gastroenterol J 2015;3:106-120. 10.1177/2050640614561668.
- Ganji A, Esmaielzadeh A, Aafzal Aghayee MA, Goshayeshi L, Ghaffarzadegan K. The clinical presentation of celiac disease: Experiences from Northeastern Iran. Middle East J Dig Dis 2014;6:93-97. PMCID: PMC4034670.
- Berry N, Basha J, Varma N, et al. Anemia in celiac disease is multifactorial in etiology: A prospective study from India. JGH Open 2018;2:196-200. 10.1002/jgh3.12073.
- Zipser RD, Patel S, Yahya KZ, Baisch DW, Monarch E. Presentations of adult celiac disease in a nationwide patient support group. Dig Dis Sci 2003;48:761-764. 10.1023/a:1022897028030.
- Lankisch PG, Martinez Schramm A, Petersen F, Dröge M, Lehnick D, Lembcke B. Diagnostic intervals for recognizing celiac disease. Z gastroenterol 1996;34:473-477. PMID: 8794542.
- Cooper BT. The delayed diagnosis of celiac disease. N Z Med J 1986;99:543-545. PMID: 3461397.
Correspondence: Wandregíselo Ponce de Leon Júnior
Email: dr.wandregastroponce@gmail.com
Acta Gastroenterol Latinoam 2021;51(4):413-419
 Revista ACTA Órgano Oficial de SAGE
Revista ACTA Órgano Oficial de SAGE