Anokhi S Kholwadwala1 ID· Eamonn M M Quigley2 ID
1 Bachelor of Science, Master of Science.
School of Engineering Medicine, Texas A&M University, Houston, Texas, USA.
2 David M. Underwood Chair of Medicine in Digestive Disorders. Co-director, Lynda K. and David M. Underwood Center for Digestive Disorders. Chief, Gastroenterology and Hepatology. Professor of Medicine, Institute of Academic Medicine, Houston Methodist Hospital. Vice-Chair for Research, Department of Medicine. Professor of Medicine, Weill Cornell Medical College. Adjunct Professor of Medicine, Texas A&M Health Sciences Center College of Medicine. Adjunct Professor, School of Medicine, University College Cork.
Acta Gastroenterol Latinoam 2025;55(4):276-291
Received: 26/11/2025 / Accepted: 16/12/2025 / Published online: 23/12/2025 / https://doi.org/10.52787/agl.v55i4.570
Summary
Small intestinal bacterial overgrowth was originally described as a cause of maldigestion and malabsorption, and its diagnosis was based on the culture of aspirates from the upper small intestine. The application of hydrogen breath tests to assess suspected small intestinal bacterial overgrowth increased the frequency of this diagnosis dramatically, and expanded its clinical context from maldigestion to include a number of non-specific gastrointestinal complaints and even some systemic ills. However, a critical review of recent literature suggests that reliance on breath tests, particularly those using lactulose as the substrate, has led to overdiagnosis of small intestinal bacterial overgrowth. Many aspects of the current concept of its diagnosis are contentious and unresolved. These include diagnostic thresholds, response to antibiotic therapy, as well as the roles of methane, hydrogen sulfide, and other gases emanating from bacterial fermentation in the gastrointestinal tract. We discuss these issues, paying particular attention to the concept of intestinal methanogen overgrowth and exploring the role of methanogens and methane in gastrointestinal homeostasis. We advocate a critical and cautious approach to the diagnosis of small intestinal bacterial overgrowth and intestinal methanogen overgrowth, and that antibiotics are prescribed are with great care. We also look forward to the application of modern molecular microbiology to the study of the small intestinal microbiome and metabolome, including a more complete understanding of methane biology.
Keywords. Small intestinal bacterial overgrowth, small intestine, malabsorption, microbiome, irritable bowel syndrome.
Sobrecrecimiento bacteriano del intestino delgado y sobrecrecimiento metanógeno intestinal: ¿están sobrediagnosticados?
Resumen
El sobrecrecimiento bacteriano del intestino delgado se describió originalmente como una causa de mala digestión y malabsorción; su diagnóstico se basaba en el cultivo de aspirados del intestino delgado superior. La aplicación de pruebas de hidrógeno en el aliento para evaluar la sospecha de sobrecrecimiento bacteriano del intestino delgado aumentó drásticamente la frecuencia de este diagnóstico y amplió su contexto clínico desde la mala digestión hasta incluir una serie de molestias gastrointestinales inespecíficas e incluso algunas enfermedades sistémicas. Sin embargo, una revisión crítica de la literatura reciente sugiere que la dependencia de las pruebas de aliento, en particular las que utilizan lactulosa como sustrato, ha llevado a un sobrediagnóstico del sobrecrecimiento bacteriano del intestino delgado. Muchos aspectos del concepto actual de su diagnóstico son controvertidos y no están resueltos. Entre ellos se incluyen los umbrales para el diagnóstico, la respuesta al tratamiento con antibióticos y el papel del metano, el sulfuro de hidrógeno y otros gases que emanan de la fermentación bacteriana en el tracto gastrointestinal. Analizamos esos temas prestando especial atención al concepto de sobrecrecimiento de metanógenos intestinales y explorando el papel de los metanógenos y el metano en la homeostasis gastrointestinal. Abogamos por un enfoque crítico y cauteloso del diagnóstico del sobrecrecimiento bacteriano del intestino delgado y el sobrecrecimiento de metanógenos intestinales, y por una gran precaución en la prescripción de antibióticos. También tenemos interés en la aplicación de la microbiología molecular moderna al estudio del microbioma y el metaboloma del intestino delgado, incluyendo una comprensión más completa de la biología del metano.
Palabras claves. Sobrecrecimiento bacteriano del intestino delgado, intestino delgado, malabsorción, microbioma, síndrome del intestino irritable.
Abbreviations
SIBO= Small intestinal bacterial overgrowth.
CFU= Colony forming units.
IMO= Intestinal methanogen overgrowth.
LBHT= Lactulose breath hydrogen test.
IBS= Irritable bowel syndrome.
GI= Gastrointestinal.
US= United States of America.
GBT= Glucose breath test.
ppm= Parts per million.
RNA= Ribonucleic acid.
DNA= Deoxyribinucleic acid.
mcrA= Alpha subunit of Methyl-coenzyme M Reductase.
CTT= Colon transit time.
SBTT= Small bowel transit time.
3S-EC-SIM= 3-stage environmental control system for intestinal microbiota.
EPR= Electron paramagnetic resonance.
mmHg= Millimeters of mercury.
FODMAP= Fermentable oligosaccharides, disaccharides, monosaccharides and polyols.
HMG CoA reductase= Hydroxymethylglutaryl coenzyme A reductase.
SHIME= Simulator of the Human Intestinal Microbial Ecosystem.
TNO TIM= TNO (Toegepast Natuurwetenschappelijk Onderzoek, Netherlands Organization for Applied Scientific Research) Intestinal Model (TIM).
HMA= Human microbiota associated.
Introduction
The idea that an abnormal, excessive, or dysfunctional bacterial population in the gastrointestinal tract can cause disease is far from new. Consider the popularity of the concept of “autointoxication” in the nineteenth century and how often colectomy was performed to rid the body of the “vile humors” emanating from “putrefaction” in the colon.1 As the importance of normal resident flora was recognized, the focus shifted to detecting abnormalities in the composition and function of microbiota, as they are now referred to, rather than flora. In the many decades that preceded the introduction of effective acid suppressants, surgery represented the optimal treatment for persistent or complicated peptic ulcer disease, and it was soon recognized that some of these procedures led to the formation of blind loops where bacteria could proliferate and impair digestion and absorption in the small intestine.2 Indeed, some of the very first descriptions of what we would now describe as small intestinal bacterial overgrowth (SIBO) involved the detection of vitamin B12 deficiency in a condition referred to as “blind loop syndrome”.3 The subsequent recognition that this same syndrome could arise in other contexts, such as colo-enteric fistulae, intestinal resection, or extensive jejunal diverticulosis necessitated a revision of terminology. The term “contaminated bowel syndrome” was adopted but it was ultimately replaced by our current term, SIBO.4 Even before metabolomics emerged, studies on hepatic encephalopathy identified the key role of bacterial metabolism of protein in the lumen of the gut in the development of the clinical manifestations of this chronic liver disease complication.5
SIBO as a cause of maldigestion and malabsorption
The latter half of the last century witnessed a series of seminal clinical research studies that described in considerable detail how the overgrowth of certain bacterial species, normally confined to the colon, could lead to maldigestion, malabsorption and even malnutrition.6, 7 Through their uptake of the vitamin, bacteria were shown to cause B12 deficiency and megaloblastic anemia, while their production of folic acid produced an unusual combination of low B12 and elevated folic acid. Deconjugation of bile acids in the proximal small intestine disrupted fat digestion leading to steatorrhea and malabsorption of fat-soluble vitamins. Direct competition between bacteria and the host for other nutrients, together with mucosal injury in more severe cases, led to hypoproteinemia, edema and even protein-losing enteropathy. Other metabolic products of bacteria and/or bacteria-host interactions could also generate symptoms, such as bacterial production of vitamin K interfering with anticoagulant efficacy and hepatic encephalopathy generated by bacterial synthesis of ammonia and other neurotoxins, as well as the generation of d-lactic acid, acetaldehyde and alcohol as a result of SIBO in certain clinical circumstances.6, 7
In this scenario, where SIBO was viewed as an important cause of the malabsorption syndrome, the likely cause of SIBO could be predicted by an understanding of those factors that maintain microbiological homeostasis in the small intestine under healthy conditions:
1. Gastric acid.
2. Intestinal and pancreatic enzymes.
3. Bile.
4. Normal intestinal motility, including the migrating motor complex.
5. An intact intestinal anatomy, including the ileocecal valve/sphincter.
6. An intact resident microbiome.
Accordingly, achlorhydria, pancreatic insufficiency, disorders such as scleroderma and chronic intestinal pseudo-obstruction, which feature impaired motility, surgical procedures that led to the formation of blind loops, resection of the ileocecal region or disease states such as jejunal diverticulosis, Crohn’s disease or radiation enteritis that led to stasis were all associated with the development of SIBO.6
In this scenario, diagnosis rested on detecting clinical and laboratory evidence of maldigestion and malabsorption, as well as defining an excess of bacteria in jejunal aspirates. Based on studies correlating bacterial counts (particularly coliforms) with laboratory measures of maldigestion/malabsorption, a threshold of 105 was proposed as the upper limit of normal for the number of colony-forming units (CFU) per ml of jejunal aspirate. For many years, this threshold reigned supreme as the standard for the diagnosis of SIBO.8 With the advent of endoscopy, sampling from the duodenum rather than the jejunum became the norm, and the threshold shifted downward to 103. However, this shift was never validated in the context of maldigestion/malabsorption. Regardless of the technique employed, aspirates were invasive, subject to contamination as the catheter traversed the upper GI and aerodigestive tracts, and likely underestimated the numbers of anaerobes.
Therefore, much effort was exerted toward developing non-invasive tests. Bile acid breath tests were first in line.9 In these tests, bile acids radiolabelled with 14C or 13C were ingested in a liquid test meal. When deconjugated by bacteria, CO2 was released, absorbed and excreted in breath samples. These tests did involve radiation exposure (when 14C was employed) and when evaluated proved inferior to aspirates. CO2 excretion following ingestion of radiolabelled D-xylose was also evaluated as a test for SIBO,9 but the arrival of hydrogen breath tests led them, by virtue of their ease of administration and measurement, to become the preferred modality for the detection of SIBO in clinical practice. Using lactulose as the substrate, hydrogen breath tests had already been widely used to measure oro-cecal transit10 and the lactulose breath hydrogen test (LBHT) was rapidly adopted as a test for SIBO in clinical practice. Yes, it was easy to perform and seemed even easier to interpret, but there was a problem that plagues the field to this day. Pandora’s box had been opened!
SIBO – beyond maldigestion and maldigestion – the era of hydrogen breath tests
About 40 years ago, the senior author of this review (EMMQ), while still a trainee, was asked to give two lectures on the pathophysiology of maldigestion and malabsorption to medical students. This was his first introduction to SIBO and, upon reading the textbooks, he marvelled at how the symptoms resulting from SIBO could be explained by bacterial disruption of various digestive processes. One moved seamlessly from risk factors to pathophysiology to symptoms and their treatment. A very neat package, indeed.
Since then, to quote the great Irish poet, William Butler Yeats, “all changed utterly”. The application of LBHT testing to individuals with irritable bowel syndrome (IBS), chronic bloating, and other GI and non-GI symptoms led to an apparent epidemic of SIBO. In the initial, seminal studies, SIBO was diagnosed in up to 84% of IBS subjects based on LBHT results. Inevitably, antibiotics, initially neomycin and later rifaximin, came to be prescribed and encouraging results were reported.11, 12 Indeed, rifaximin ultimately came to be approved in the US for non-constipated IBS,13 but it is important to note that in the pivotal, phase III study on which this approval was based, breath testing was performed in only a small minority of subjects at baseline,14 so this was a study on non-constipated IBS, not on SIBO in IBS. It should also be noted that the therapeutic gain for rifaximin in IBS was small, around 7 – 10%.13 So, to again paraphrase Yeats, had “a terrible beauty been born”? Was SIBO the main cause of IBS or was something else going on?
Several factors led to the almost universal adoption of hydrogen breath tests for the diagnosis of SIBO in the United States. Of these, ease of performance and acceptability to the patient were foremost. The test could be readily performed and interpreted in the gastroenterologist’s office. Over the subsequent decades studies employing lactulose as the substrate demonstrated apparently high prevalence rates for SIBO, not only in IBS, but across a wide range of gastrointestinal and systemic disorders. Notes of caution were evident at an early stage in the history of the lactulose breath hydrogen test – its origins, after all, lay in the assessment of oro-cecal transit. Concerns regarding the impact of accelerated small intestinal transit, such as may occur in diarrheal states and potentially exacerbated by lactulose itself, on the interpretation of this test were raised and borne out in studies that simultaneously tracked small bowel transit and the breath hydrogen peak.15 It soon became evident that rapid transit led to a false positive test. Comparisons with simultaneously performed small bowel aspirates and glucose breath tests further undermined the reliability of the lactulose breath hydrogen test and made it abundantly clear that the very high rates of SIBO reported with this test included a very significant burden of test artifact.16, 17 In one study, the false positive rate was so high that it failed to differentiate IBS from controls.17 It should come as no surprise, therefore, that a multinational panel of experts recently recommended the abandonment of the lactulose breath hydrogen test in the diagnosis of SIBO.18
The glucose breath hydrogen test and small intestinal aspirates have their own set of problems. Prevalence rates for SIBO are certainly lower when glucose is used as the substrate for a hydrogen breath test, and positive tests are more frequent among IBS subjects than among controls,17 but false positive tests do occur, albeit at a much lower rate than when lactulose is the substrate. For example, Lin and Massey reported a 45% false positive rate for glucose breath tests due to rapid transit.19 For aspirates, whether obtained from the jejunum or duodenum, contamination with microbes in the upper aerodigestive tract and protection of strict anaerobes from exposure to oxygen can be a problem. Innovations in catheter design have minimized the impact of contamination but preservation of anaerobes remains an issue.20, 21
At least two meta-analyses have examined the outcomes of diagnostic tests for SIBO among subjects with IBS. Both found that SIBO prevalence in IBS is highly dependent on test modality, with the highest rates being noted with a lactulose breath test and the lowest with aspirates that define SIBO at a threshold of 103.16, 17 In the meta-analysis performed by Shah and colleagues, heterogeneity was extremely high for all tests, with the exception of the glucose breath test. This undoubtedly contributed to their finding that SIBO prevalence was no different from that in the control group when using the LBHT or aspirates and cultures interpreted based on a cut-off of threshold of 105.17 (Table 1).
Table 1. Comparison of the results of various tests for small intestinal bacterial overgrowth (SIBO) among subjects with irritable bowel syndrome (IBS). Based on Shah et al 17
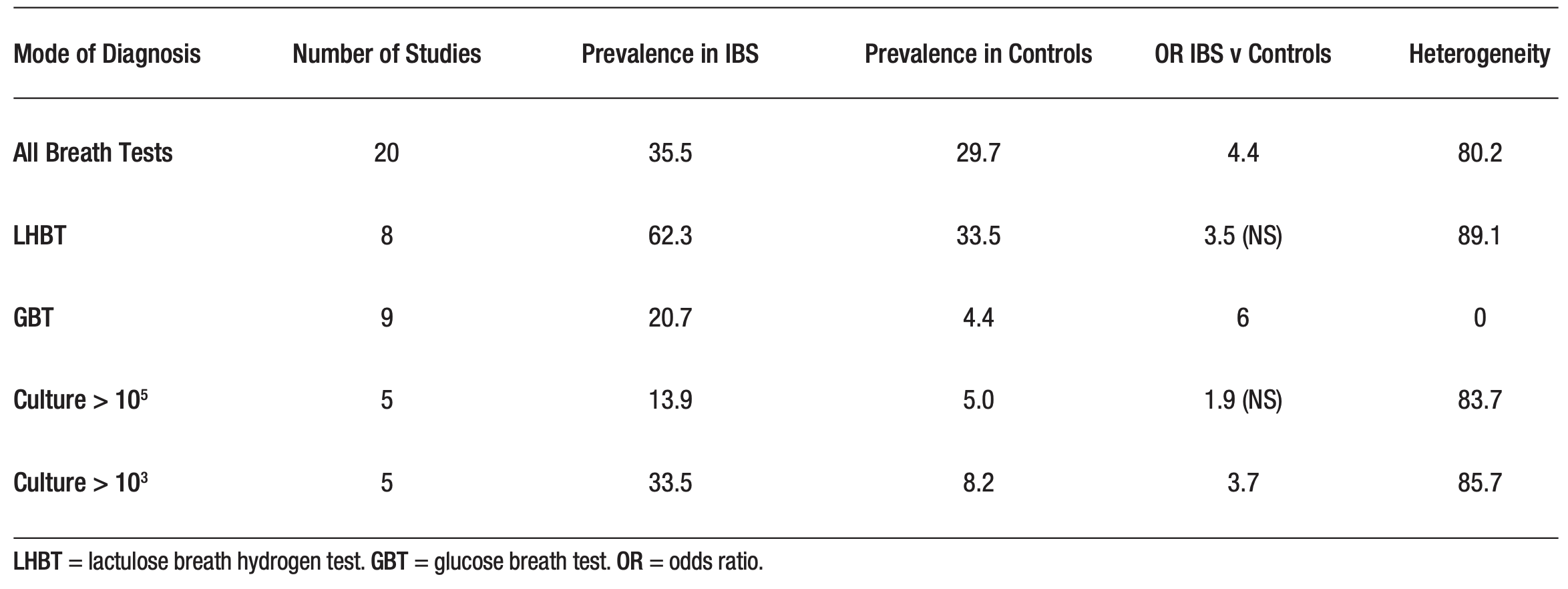
The diagnostic thresholds for all of these tests remain contentious, and variations in their definition are a major contributor to the uncertainty surrounding the interpretation of these tests. Should the cut-off for an aspirate be 103 or 105? The latter, though time honoured, was developed on the basis of aspirates taken from the proximal jejunum. Given the luminal conditions extant in the duodenum, a lower threshold seems logical and has been widely employed, but has it been validated? Studies comparing aspirates and microbiome populations (evaluated by high-throughput sequencing) in the lumen or at the mucosal surface have provided conflicting results, with some demonstrating a high degree of correlation between aspirates and an altered microbiome and others failing to demonstrate any significant relationship.22-28 Turning to hydrogen breath tests, two issues need to be addressed: At what time after substrate ingestion is a peak regarded as “early”, and what rise in parts per million (ppm) should be regarded as significant? Various intervals have been proposed with the North American Consensus on Hydrogen and Methane-Based Breath Testing in Gastrointestinal Disorders recommending that peaks noted before 90 minutes have elapsed should be regarded as reflecting bacterial fermentation within the small intestine.29 Given that oro-cecal transit rates as rapid as 48 ± 6 minutes have been reported with a 10 g dose of lactulose30 when administered as a drink (without a meal), and that others have noted a mean oro-cecal transit time of 75 minutes with 80 g of glucose,19 the problems with a 90-minute cut-off become glaringly obvious, as revealed in a recent study.31 With regard to what degree of rise in breath hydrogen from baseline is considered significant, the North American Consensus defined a rise of ≥ 20 ppm as indicative of a positive test;29 a threshold that seems to accord with clinical features but has not been rigorously tested in terms of predictability for therapeutic response. Criteria for a significant rise in methane will be discussed later in this review.
The clinician will ask the critical question – do any of these test parameters predict therapeutic response? Here we have surprisingly limited data and what is available is not of the highest quality. Various antibiotic regimens have been advocated over the years, but few have been subjected to the rigor of adequately powered, randomized placebo-controlled clinical trials. The limitations of the available literature on antibiotic therapy in SIBO were laid bare in a recent meta-analysis that concluded that while antibiotics appeared to be efficacious in treating SIBO, small sample sizes and poor data quality limited this interpretation.32 They also noted that symptomatic response rates to antibiotics in IBS appeared to be higher in IBS patients with SIBO.32 As mentioned above, a small number of subjects enrolled in the pivotal trial that led to the approval of rifaximin in non-constipated IBS in the US underwent breath testing before and after treatment with rifaximin.14 While a positive baseline LBHT was predictive of a better response to rifaximin, and three-quarters of those who responded to rifaximin had switched from a positive to a negative breath test after treatment, 56% of those who remained positive after treatment were also designated as responders.14 Large multicenter studies of antibiotic efficacy in SIBO related to IBS are clearly needed. Another major issue for the clinician is recurrence. One study reported a recurrence rate of just under 44% (as indicated by glucose breath test positivity) following SIBO antibiotic therapy.33 While rifaximin, perhaps the most widely prescribed antibiotic for SIBO, has been shown to retain efficacy on repeat therapy34 clinicians must be aware of the specter of antibiotic resistance, which is a global public health concern. Although rifaximin has traditionally been considered low-risk for the induction of antibiotic resistance,35 recent reports on the emergence of resistance to important human pathogens among individuals on long-term rifaximin therapy for hepatic encephalopathy36, 37 are, therefore, concerning.
First, the clinician must be absolutely sure that SIBO is present and responsible for the patient’s symptoms. Second, the clinician must be sure that persistent or recurrent symptoms are still attributable to this diagnosis. Otherwise, repeated courses of broad-spectrum antibiotics will be prescribed to address non-specific symptoms and/or assuage patients convinced that they have SIBO and need another round of antibiotics, with deleterious outcomes for the patient and the community. As clinicians, it is our responsibility to accurately diagnose SIBO and embark on treatment only when convinced that SIBO is really evident and relevant to the patient’s symptoms and clinical context. This is clearly much easier in the setting of maldigestion and malabsorption but challenging in a scenario where neither is evident and symptoms are non-specific.
Intestinal Methanogen Overgrowth – Methane is added to the equation
Most commercially available breath testing equipment now provides a readout of methane levels, in addition to hydrogen levels in breath samples. This led to a dramatic increase in the reporting of what is referred to as intestinal methanogen overgrowth (IMO). Before we discuss the diagnosis and management of IMO let us review what we know of the biology of methane.
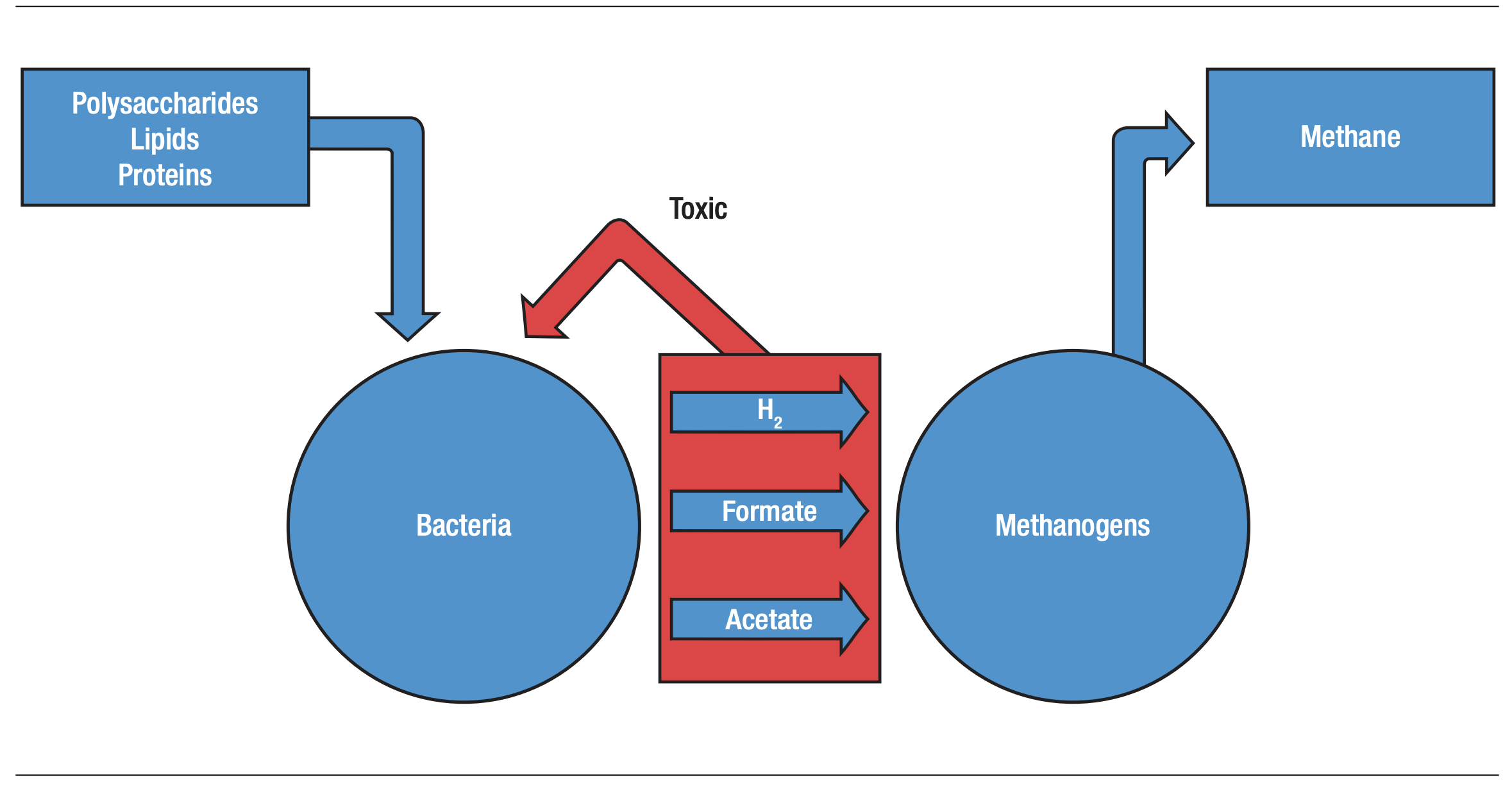
The appearance of methane in human breath derives from the actions of a group of Archaeal microorganisms referred to as methanogens in the human gastrointestinal tract. By combining carbon (from carbon dioxide) with hydrogen, methanogens such as Methanobrevibacter smithii produce methane. Since four molecules of hydrogen are utilized to produce one molecule of methane, this reaction not only reduces the concentration of hydrogen but also the total intraluminal gas volume38, 39 (Figures 1 and 2).
Figure 1. Methane Biosynthesis in the human gastrointestinal tract
Although proportions vary, largely due to sampling technique, it is estimated that approximately one-third of the Western population produces methane,39 and this proportion has remained relatively stable over the last several decades.40
Methane production is influenced by both genetic and environmental factors.39 Early twin studies with breath samples of 274 twin pairs found that methane excretion was attributed more to shared and unique environmental effects than to genetic effects.41 Environmental factors include low-fat intake and low vitamin B12 intake, which increase methanogen populations.42
However, a more recent study of methanogen carriage, measured using the mcrA gene as a marker, showed a concordance rate of 74% in monozygotic twins versus 15% in dizygotic twins, indicating a strong host genetic influence on the presence of gut methanogens.43
Archaea and Methanogens
Archaea are microorganisms that form a domain distinct from bacteria. Although similar in size and general morphology to bacteria, distinguishing features include the ability to perform methanogenesis and the absence of peptidoglycan in the cell wall. They also have a more complex RNA polymerase which is similar to that of eukaryotes.44 Methanogens are one group of the domain Archaea and are strict anaerobes.44
The presence of methanogens is influenced by several factors, some of which are undoubtedly interrelated: stool consistency, bile acid concentrations, oxygen tension, and proximity to bacterial populations.44 Methanogens are most common in areas of the GI tract where stool is more solid, notably the left colon.39, 40
Normally, only a small amount of primary bile acids gains access to the colon, where they undergo biotransformation to secondary bile acids through interaction with gut microbiota.45 These primary and secondary bile acids, particularly in their lipophilic, unconjugated forms, act as detergents and disrupt cell membranes.45 This leads to bacteriostatic and bactericidal effects on susceptible bacteria.46 However, certain bacterial species, including Salmonella, E. coli, and Campylobacter, exhibit tolerance to bile acids.45
Methanogens, on the other hand, are poorly tolerant. Exposure to higher concentrations of bile acids, particularly unconjugated ones, results in a parallel decline in methane production and an increase in hydrogen concentration.47
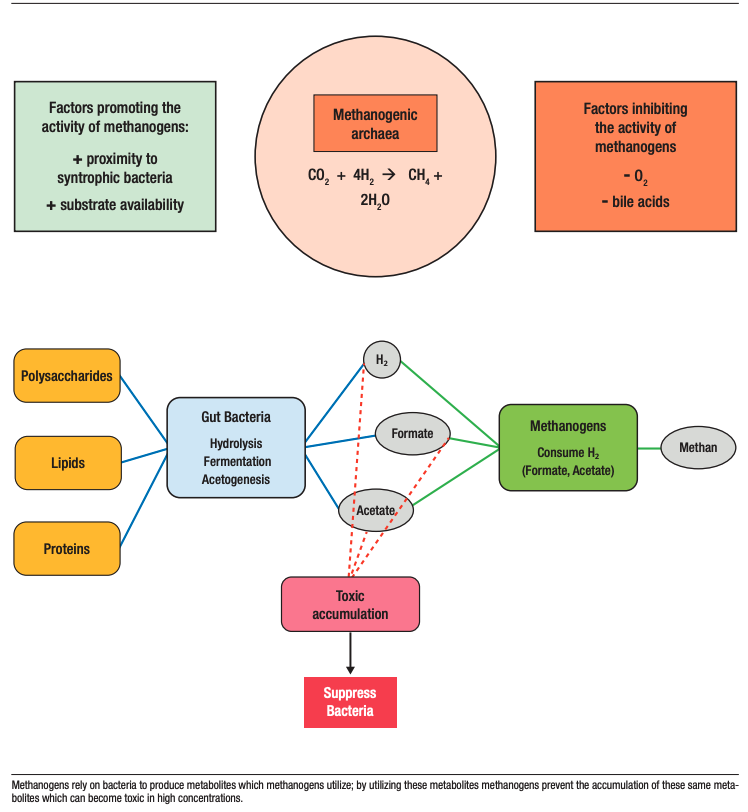
Methanogens can only survive in an environment where they can maintain intimate contact with certain bacteria, as they need these bacteria to process polymers into usable substrates (Figure 2).
Polysaccharides, lipids, and proteins all need to undergo hydrolysis, fermentation, and acetogenesis before they can be used by methanogens. Benefits are mutual.
The aforementioned end products of bacterial metabolism, such as hydrogen, formate, and acetate, can become toxic to the bacteria through acidification of the milieu (Figures 1 and 2); however, methanogens can minimize their accumulation.48 Factors promoting these syntropic relationships between bacteria and methanogens include short interspecies distances for metabolite transfer (thus the relationship to a more solid stool), as well as the presence of bacteria that, like methanogens, are slow-growing and can provide a predictable supply of the metabolites required by methanogens.48
Examples of such bacteria include the genera Christensenella and Bacteroides.48 This interaction between methanogens and bacteria can disrupt the microbiome in one of two ways48 (Figure 2):
- An increased methanogen population requires bacteria to generate more products for methanogen metabolism, thereby altering bacterial populations, or,
- A decreased methanogen population will be unable to create an environment capable of supporting appropriate bacteria due to the accumulation of toxic metabolites.
However, current evidence does not support classifying methanogens as pathogens or pathobionts, and there is no definitive proof that their absence or presence directly causes disease.48 It seems more likely that methanogens may influence health via their interactions with bacteria and their role in stabilizing the microbiome.48
Due to this relationship between bacteria and methanogens, a potential role for methanogens as probiotics has been suggested.48
Figure 2. Syntropic relationships between bacteria and methanogens
Methane and Constipation
In clinical medicine, much of the interest around methane and methanogens relates to a proposed role for methane in the pathogenesis of constipation. One of the first studies to describe an association between methane and bowel function was a report of an association between elevated levels of methane in the breath and encopresis (but not constipation) in children, with methane being present in the breath samples of 15% of the control children and 65% of encopretic children.49
Subsequently, a study of 551 subjects, 296 of whom had IBS, found that the severity of constipation symptoms (evaluated via a questionnaire) correlate with methane excretion on a lactulose breath test.50 Additionally, a study of 87 IBS patients found that greater methane production on a lactulose breath test correlated with increased constipation severity, fewer bowel movements per day, and harder stool.51
Recently, a systematic review and meta-analysis of 19 studies, encompassing 1,293 individuals with IMO and 3,208 controls, found that constipation occurred in 47% of IMO patients in comparison to 38% of controls. While this association held true regardless of whether glucose or lactulose was used as the substrate for the breath test or the study was prospective or retrospective, there were limitations – not every study used verified symptom questionnaires, the diagnosis for IMO was not standardized, and not all breath tests were performed according to guidelines. Importantly, there was also significant heterogeneity between studies.52
These findings beg the question: Is methane the cause of constipation or a mere consequence?
Impacts of methane on intestinal function
There are limited data on the impact of methane on intestinal motility or secretion, which are pivotal factors to the pathogenesis of constipation. One study evaluated the impact of methane infused into the small intestine of dogs on intestinal transit time; compared to an infusion of room air, methane slowed small intestinal transit by 59%.53 In other components of this work, exposure of guinea pig ileum to methane gas increased contractile activity, and a retrospective review noted that methane-producing IBS subjects had a higher small bowel motility index (as measured by small bowel manometry) than those who produced hydrogen. To explain the seemingly contradictory results from the dog experiments and the guinea pig and human studies, the authors suggested that methane promoted non-propulsive contractions and thereby impaired transit.53
Previously, Jahng and colleagues noted that methane delayed ileal peristaltic conduction velocity by augmenting contractility in guinea pigs.54 Hydrogen shortened colonic transit, and that effect was more prominent in the proximal colon than the distal colon. While these studies demonstrate an effect of methane on small intestinal motility, it is unlikely that such an effect contributes to constipation given the modest contribution of small intestinal transit time to the time it takes to transit from the mouth to the anus.
A small number of studies have examined relationships between constipation and gastrointestinal transit in humans. In one study, 96 patients with chronic constipation and 106 control subjects underwent measurements of colonic transit time using the radio-opaque marker technique and glucose breath tests to define IMO. Patients with chronic constipation were more likely to be methane producers, and within this group, those with slower transit times were more likely to exhale methane. A methane concentration of ≥ 3 ppm was present in breath testing of 75% of constipated patients with slow transit time, 44% of constipated patients with normal transit time, and 28% of non-constipated controls.55
Talamantes and colleagues compared breath test results (using either lactulose or glucose as substrate), transit times and intraluminal pH values (measured by a wireless motility capsule) along the gastrointestinal tract among 196 subjects. They found that colonic transit time (CTT) was significantly longer in patients with breath tests that were positive on hydrogen and/or methane criteria (40 h : 29 min) compared to those with negative breath tests (28 h:51min). In those with IMO, small bowel transit time (SBTT) was somewhat delayed (5h : 15 min vs 4 hr : 32 min) and CTT prolonged (44 hr : 23 min vs 28 hr : 51 min) compared to patients with negative results.56 This also suggests that the main effect of methane in relation to constipation relates to impacts on the colon, not the small intestine. These studies do not, of course, determine whether methane is a consequence or a cause of constipation.
Constipation as a cause of methane production
How does gut transit influence the composition of the microbiome? Many factors influence the wide inter-subject variability in transit, including host sex, age, intestinal length, and dietary considerations such as carbohydrate, fat, and fiber intake. However, it is also evident that there is a bidirectional relationship between transit time and the fecal microbiome.57 For example, longer colonic transit times alter the luminal milieu by increasing pH in the distal colon, decreasing carbohydrate availability, increasing proteolytic bacterial metabolism, and enhancing microbial conversion and reabsorption of bile acids.57 Longer transit times, including slower stool movement, more solid stool, and altered bile acid concentrations, create an environment favorable for methanogen proliferation.57
The effects of transit on the microbiome were demonstrated in studies using the 3-stage Environmental Control System for Intestinal Microbiota (3S-ECSIM) in vitro system. Prolonging transit times from
48 hours to 96 hours promoted a bloom of Clostridium cluster XIV and potentially favorable conditions for the growth of methanogens, although archaea were not enumerated in this study.58
Similar effects have been demonstrated in humans; when transit was pharmacologically delayed by loperamide, methanogen counts in fecal samples increased, and concentrations of methane in the breath were higher. In contrast, the prokinetic agent cisapride was shown to decrease methanogen counts.59
Intestinal Methanogen Overgrowth (IMO) – diagnostic issues
Relationships between breath methane levels and methanogen populations are complex. For example, it has been shown that the absence of methane on breath test should not be interpreted as reflecting an absence of methanogens in the GI tract; indeed, Methanobrevibacter smithii has been detected in the stool of those with positive and negative lactulose breath tests.60 Moreover, a population density of at least 4.2 × 105 copies per gram of stool, or 1.2% of total gut bacteria, is needed to produce a breath test positive for methane.60 Aditionally, Di Stefano and colleagues found that the correlation between rectal and breath levels of methane was poor.61
Various criteria for breath test positivity in relation to methane have been proposed, contributing to uncertainty regarding the percentage of the population that are methane producers39 and, in the clinical arena, on the diagnosis of what is referred to as IMO. The North American Consensus recommended that a breath methane level of ≥ 10 ppm above baseline at any time after substrate (either lactulose or glucose) ingestion was indicative of small intestinal methanogen overgrowth.29 The clinical relevance of this threshold will ultimately be validated by its ability to guide successful therapy.
Do interventions that suppress methanogens have a clinical impact?
To date, unfortunately, there have been few high-quality trials of any intervention targeting methanogens or methane production. Some data exist regarding the use of dietary interventions to modulate methanogen populations. One retrospective review assessed the impact of an elemental diet on 93 patients with IBS. After 21 days on this diet, 85% had normalized their lactulose breath tests and symptoms such as diarrhea and constipation had improved in parallel.62 Similar effects were reported in a case study.63 Additionally, a response to a low-FODMAP diet has been linked to higher levels of fecal methanogens at baseline.64 Whether this diet reduces methanogen populations or breath hydrogen levels has not been established.
In relation to treatment, some antibiotics have been shown to be effective against methanogens in both in vitro sensitivity testing and in vivo studies.65 In vitro, M. smithii strains displayed strong resistance to ampicillin, streptomycin, gentamicin, rifampicin, ofloxacin, tetracycline, amphotericin B, and vancomycin, moderate resistance to chloramphenicol, and susceptibility to bacitracin, metronidazole, ornidazole, and squalamine.65 Due to differences in bacterial, fungal and archaeal structure, methanogens were resistant to agents that target bacterial cell walls (ampicillin, vancomycin) or ergosterol in fungal cell membranes (amphotericin B). Additionally, because archaeal RNA polymerases are more related to eukaryotic RNA polymerases than bacterial, methanogens are resistant to rifampicin which inhibits bacterial RNA polymerase. The resistance to ofloxacin can be explained via a weaker drug interaction with the archaeal equivalent of DNA gyrase, as well as efflux pumps encoded by methanogens. Finally, methanogens possess genes that make them resistant to chloramphenicol, streptomycin, and gentamicin. As for the antimicrobials effective against methanogens, metronidazole can damage methanogen DNA, bacitracin can interrupt pseudomurein in the cell walls of M. smithii, and squalamine may depolarize organisms.65
Turning to clinical studies, a subanalysis of a double-blind randomized controlled trial focusing on patients with IBS-C and a positive methane breath test result showed that 44% of those who received neomycin experienced an improvement in constipation compared to only 5% of those administered a placebo.66 The beneficial effects of antibiotics in addressing methanogens and improving constipation have also been demonstrated in case reports.67 Similar benefits have been demonstrated for the probiotic Lactobacillus reuteri.68, 69
Recently, statins have been explored as a therapeutic strategy. Statins (especially lactone statins) are taken up by Methanobrevibacter smithii, where they inhibit the biosynthesis of archaeal cell membranes by targeting HMG-CoA reductase. An advantage of statins over antibiotics is that statins only target archaea, leaving bacterial communities untouched.70 One in vitro study verified this effect, and showed that increasing doses of lovastatin resulted in lower methanogen growth and methane production.71
Methanogens in the Small Intestine
When comparing the environments of the small intestine and the large intestine, factors such as flow rate, bile acid concentration, and oxygen tension are important considerations for the survival and proliferation of methanogens.
The colon can support obligate anaerobic bacteria and methanogens due to its high microbial density, the availability of carbohydrates that humans cannot digest, low oxygen tension, and low concentrations of primary bile acids.72 In contrast, the small intestine has an overall low microbial density due to high flow rate and secretion of bile acids.73 Additionally, host absorption of easily digestible carbohydrates in the proximal small intestine will limit access for microbes to these resources.74 Oxygen tension along the gut is one of the most important factors determining the survival and growth of obligate anaerobes such as methanogens. In the GI tract, both longitudinal (along the length of the intestine from mouth to anus) and transversal (from lumen to mucosa) oxygen gradients are evident. However, reported values for these gradients vary widely depending on the measurement method, sampling location (luminal vs. juxtamucosal), and species studied.75 A longitudinal gradient from 58 mmHg in the stomach to 32 mmHg in the duodenum and 3 mmHg in the sigmoid colon was demonstrated in mice using spectral-spatial electron paramagnetic resonance (EPR) imaging to measure luminal pO2.76 In terms of a radial gradient, EPR oximetry in rats showed that oxygen tension in the small intestine is highest in the intestinal wall (~54.5 mm Hg) and lowest in the lumen (~9.6 mm Hg).77 Again, reported values vary widely, but regardless of the exact numbers, a radial gradient is well supported.78-80 This potentially creates areas in the small intestine where methanogens could survive.81 Nonetheless, given the much less hospitable environment of the small intestine, can methanogens even exist there?
The limited available data on methanogens in the small intestine does not provide a clear answer. An analysis of gas composition in various sections of the GI tract in slaughtered pigs failed to detect methane in the stomach or small intestine, and detected minimal levels in the proximal colon and cecum and higher levels in the distal colon.82 In contrast, very recent studies by Villanueva-Millan and colleagues support the possibility that methanogens are present in the proximal small intestine. Using shotgun metagenomics, they detected methanogens and hydrogen sulfide-producing bacteria in the human duodenum. They also correlated their presence with the detection of methane and hydrogen sulfide, respectively, in the breath.81 These findings are suggestive, but not conclusive, as methanogen viability was not defined. In the absence of such evidence, it is possible that non-viable methanogens passaging through from the oropharynx were being detected.
Difficulties in Studying the Small Intestine – How New Technologies May Move Us Forward
Challenges with access, in particular, and with retrieving representative microbiome samples have limited our ability to study the small intestine and our understanding of the human small intestinal microbiome in health or disease.83
Due to these difficulties, various systems have been developed in an attempt to model the human small intestine. In vitro options have included the Simulator of the Human Intestinal Microbial Ecosystem (SHIME) model84 and TNO (Toegepast Natuurwetenschappelijk Onderzoek, Netherlands Organization for Applied Scientific Research) Intestinal Model (TIM).85 While these systems can simulate luminal conditions in the small intestine, there are limitations, including the absence of an epithelial or mucosal layer, a microbiome that may not be representative of the complete small intestinal microbiome, and the near impossibility of recreating the complexities of intestinal motor activity. Additionally, validation is challenging, as the human small intestine cannot be readily examined for comparison.83
In vivo, pigs are the best animal model to represent human small intestinal physiology. Like humans, pigs are omnivores and share comparable dietary needs. Their gastrointestinal systems exhibit similar gene regulation, transit times, structural features, nutrient absorption processes, and a gut microbiota predominantly composed of Firmicutes and Bacteroidetes. Additionally, the mucosal barrier in pigs closely resembles that in humans, contributing to shared vulnerability to certain pathogens. The relevance of human-microbiota-associated (HMA) gnotobiotic pigs, which are colonized with human gut microbes, for studying human gastrointestinal health, is further enhanced.86 In fact, these HMA pigs have been commonly used to study probiotics, prebiotics, and immune responses.87
Until recently, samples from human subjects have been derived exclusively from the most proximal and distal reaches of the small intestine. The challenges of duodenal and proximal jejunal sampling have already been discussed. At the other end of the small intestine, samples have been obtained from the distal regions using various methods, including naso-intestinal tubes,88 colonoscopic intubation of the distal ileum to obtain biopsies or aspirates,89 and collection of effluent from patients with ileostomies.90 All of these approaches have limitations.
Innovations in capsule technology may soon permit sampling along the length of the small intestine. On the one hand, capsules have been developed that can provide “live” readouts of luminal gas concentrations as the capsule transits the gastrointestinal tract,91-94 thus providing insights into one aspect of microbial metabolism. On the other hand, systems are already available that can sample intraluminal contents at pre-defined locations along the small intestine and colon.95 These capsules incorporate a collection bladder and a one-way valve that allows for sample collection (400 µl of luminal contents) after opening in an environment with a certain pH.95 These capsules were used in a study of 15 healthy individuals who each ingested 4 capsules designed to dissolve at a specific location along the intestinal tract. Simultaneous sampling of stool and saliva revealed the inadequacies of stool and saliva samples in reflecting the small intestinal microbiome. A longitudinal gradient of microbiota and metabolites was evident, and distinct microbial communities and metabolic profiles were noted in different gut regions. As expected, bile acid profiles varied along the gut and correlated with the presence of specific microbial populations.95 Further refinements in capsule technology may soon provide a comprehensive description of the human small intestinal microbiome and metabolome and provide the basis for studies in disease states with an accompanying revision of the definitions of SIBO and IMO.
Conclusions
SIBO has witnessed a dramatic shift in its scope in recent years, evolving from a relatively uncommon cause of malabsorption and maldigestion among individuals with a surgically modified or diseased intestine, to being implicated in the pathogenesis of common chronic digestive and systemic complaints. Along the way, technical advances have permitted the sampling of not just hydrogen but also methane and hydrogen sulfide in the breath, leading to the introduction of new concepts such as IMO. The use of lactulose as the substrate for hydrogen breath tests generated a high rate of false positive tests, leading some to suggest that it should be abandoned as a test for SIBO or IMO. Glucose breath tests are more reliable but also susceptible to some artifact. Meanwhile, the development of catheters that reduce the likelihood of contamination from oropharyngeal or gastric microbes and protect anaerobes have regenerated enthusiasm for aspiration of small intestinal contents. This approach will prosper in research but it is unlikely to enter routine medical practice. Novel capsule technologies promise to provide non-invasive yet detailed analyses of the small intestinal microbiome and metabolome. A review of the biology of methane production and the physiological effects of methane in the gastrointestinal tract reveals considerable complexity and some uncertainty. Whether methane is generated to a physiologically relevant extent in the small intestine is unclear and the relationship between methane and constipation (whether it is a cause or a consequence) continues to be debated.
Intellectual property. The authors declare that the data presented in the manuscript are original and were carried out at their belonging institutions.
Funding. Supported, in part, by the Josephine Hughes Sterling Foundation and the Lynda K and David M Underwood Center for Digestive Health.
Conflict of interest. EMMQ is a consultant to Atmo Biosciences.
Copyright
© 2025 Acta Gastroenterológica latinoamericana. This is an open-access article released under the terms of the Creative Commons Attribution (CC BY-NC-SA 4.0) license, which allows non-commercial use, distribution, and reproduction, provided the original author and source are acknowledged.
Cite this article as: Kholwadwala A S and Quigley E M M. Small Intestinal Bacterial Overgrowth and Intestinal Methanogen Overgrowth: Are they Overdiagnosed?. Acta Gastroenterol Latinoam. 2025;55(4):276-291. https://doi.org/10.52787/agl.v55i4.570
References
- Chen TS, Chen PS. Intestinal autointoxication: a medical leitmotif. J Clin Gastroenterol. 1989;11:434-41.
- Card WI. Blind loop syndrome. Proc R Soc Med. 1959;52:28-31.
- Barker WH, Hummel LE. Macrocytic anemia in association with intestinal strictures and anastomosis – review of the literature and report of two new cases. Bull Johns Hopkins Hosp. 1939;64:215-56.
- Gracey M. Intestinal absorption in the “contaminated small-bowel syndrome”. Gut. 1971;12:403-10.
- Sherlock S, Summerskill WH, White LP, Phear EA. Portal-systemic encephalopathy; neurological complications of liver disease. Lancet. 1954;267:454-7.
- Bushyhead D, Quigley EMM. Small Intestinal Bacterial Overgrowth-Pathophysiology and Its Implications for Definition and Management. Gastroenterology. 2022;163:593-607.
- Quigley EM. Small intestinal bacterial overgrowth: from malabsorption to misinterpretation. Minerva Gastroenterol (Torino). 2025;71:65-73.
- Tabaqchali S, Okubadejo OA, Neale G, Booth CC. Influence of abnormal bacterial flora on small intestinal function. Proc R Soc Med. 1966;59:1244-6.
- King CE, Toskes PP, Guilarte TR, Lorenz E, Welkos SL. Comparison of the one-gram d-[14C]xylose breath test to the [14C]bile acid breath test in patients with small-intestine bacterial overgrowth. Dig Dis Sci. 1980;25:53-8.
- Bond JH, Levitt MD, Prentiss R. Investigation of small bowel transit time in man utilizing pulmonary hydrogen (H2) measurements. J Lab Clin Med. 1975;85:546-55.
- Pimentel M, Chow EJ, Lin HC. Eradication of small intestinal bacterial overgrowth reduces symptoms of irritable bowel syndrome. Am J Gastroenterol. 2000;95:3503-6.
- Pimentel M, Chow EJ, Lin HC. Normalization of lactulose breath testing correlates with symptom improvement in irritable bowel syndrome. a double-blind, randomized, placebo-controlled study. Am J Gastroenterol. 2003;98:412-9.
- Pimentel M, Lembo A, Chey WD, Zakko S, Ringel Y, Yu J, et al. Rifaximin therapy for patients with irritable bowel syndrome without constipation. N Engl J Med. 2011;364:22-32.
- Rezaie A, Heimanson Z, McCallum R, Pimentel M. Lactulose Breath Testing as a Predictor of Response to Rifaximin in Patients With Irritable Bowel Syndrome With Diarrhea. Am J Gastroenterol. 2019;114:1886-93.
- Yu D, Cheeseman F, Vanner S. Combined oro-caecal scintigraphy and lactulose hydrogen breath testing demonstrate that breath testing detects oro-caecal transit, not small intestinal bacterial overgrowth in patients with IBS. Gut. 2011;60:334-40.
- Ford AC, Spiegel BMR, Talley NJ, Moayyedi P. Small intestinal bacterial overgrowth in irritable bowel syndrome: systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2009;7:1279-86.
- Shah A, Talley NJ, Jones M, Kendall BJ, Koloski N, Walker MM, et al. Small Intestinal Bacterial Overgrowth in Irritable Bowel Syndrome: A Systematic Review and Meta-Analysis of Case-Control Studies. Am J Gastroenterol. 2020;115:190-201.
- Kashyap P, Moayyedi P, Quigley EMM, Simren M, Vanner S. Critical appraisal of the SIBO hypothesis and breath testing: A clinical practice update endorsed by the European society of neurogastroenterology and motility (ESNM) and the American neurogastroenterology and motility society (ANMS). Neurogastroenterol Motil. 2024;36:e14817.
- Lin EC, Massey BT. Scintigraphy Demonstrates High Rate of False-positive Results From Glucose Breath Tests for Small Bowel Bacterial Overgrowth. Clin Gastroenterol Hepatol. 2016;
14:203-8. - Shanahan ER, Zhong L, Talley NJ, Morrison M, Holtmann G. Characterisation of the gastrointestinal mucosa-associated microbiota: a novel technique to prevent cross-contamination during endoscopic procedures. Aliment Pharmacol Ther. 2016;43:1186-96.
- Karunaratne TB, Sharma A, Rao SSC. Small-bowel aspiration during upper esophagogastroduodenoscopy: Rao technique. VideoGIE. 2021;6:152-4.
- Li J, Zhang R, Ma J, Tang S, Li Y, Li Y, et al. Mucosa-Associated Microbial Profile Is Altered in Small Intestinal Bacterial Overgrowth. Front Microbiol. 2021;12:710940.
- Shin AS, Gao X, Bohm M, Lin H, Gupta A, Nelson DE, et al. Characterization of Proximal Small Intestinal Microbiota in Patients With Suspected Small Intestinal Bacterial Overgrowth: A Cross-Sectional Study. Clin Transl Gastroenterol. 2019;10:e00073.
- Saffouri GB, Shields-Cutler RR, Chen J, Yang Y, Lekatz HR, Hale VL, et al. Small intestinal microbial dysbiosis underlies symptoms associated with functional gastrointestinal disorders. Nat Commun. 2019;10:2012.
- Leite G, Morales W, Weitsman S, Celly S, Parodi G, Mathur R, et al. The duodenal microbiome is altered in small intestinal bacterial overgrowth. PLoS One. 2020;15:e0234906.
- Barlow JT, Leite G, Romano AE, Sedighi R, Chang C, Celly S, et al. Quantitative sequencing clarifies the role of disruptor taxa, oral microbiota, and strict anaerobes in the human small-intestine microbiome. Microbiome. 2021;9:214.
- Bamba S, Imai T, Sasaki M, Ohno M, Yoshida S, Nishida A, et al. Altered gut microbiota in patients with small intestinal bacterial overgrowth. J Gastroenterol Hepatol. 2023;38:61-9.
- Leite G, Rezaie A, Mathur R, Barlow GM, Rashid M, Hosseini A, et al. Defining Small Intestinal Bacterial Overgrowth by Culture and High Throughput Sequencing. Clin Gastroenterol Hepatol. 2024;22:259-70.
- Rezaie A, Buresi M, Lembo A, Lin H, McCallum R, Rao S, et al. Hydrogen and Methane-Based Breath Testing in Gastrointestinal Disorders: The North American Consensus. The American Journal of Gastroenterology. 2017;112:775.
- Read NW, Miles CA, Fisher D, Holgate AM, Kime ND, Mitchell MA, et al. Transit of a meal through the stomach, small intestine, and colon in normal subjects and its role in the pathogenesis of diarrhea. Gastroenterology. 1980;79:1276-82.
- Jiménez-Castillo RA, Félix-Téllez FA, Vargas-Basurto JL, Noriega-García NB, Suárez-Fernández M, García-Zermeño KR, et al. Diagnostic Reliability of Glucose and Lactulose Breath Tests: Insights From a Test-Retest Study in Healthy Adults. Neurogastroenterol Motil. 2025;e70089.
- Takakura W, Rezaie A, Chey WD, Wang J, Pimentel M. Symptomatic Response to Antibiotics in Patients With Small Intestinal Bacterial Overgrowth: A Systematic Review and Meta-analysis. J Neurogastroenterol Motil. 2024;30:7-16.
- Lauritano EC, Gabrielli M, Scarpellini E, Lupascu A, Novi M, Sottili S, et al. Small intestinal bacterial overgrowth recurrence after antibiotic therapy. Am J Gastroenterol. 2008;103:2031-5.
- Lembo A, Pimentel M, Rao SS, Schoenfeld P, Cash B, Weinstock LB, et al. Repeat Treatment With Rifaximin Is Safe and Effective in Patients With Diarrhea-Predominant Irritable Bowel Syndrome. Gastroenterology. 2016;151:1113-21.
- DuPont HL. The potential for development of clinically relevant microbial resistance to rifaximin-α: a narrative review. Clin Microbiol Rev. 2023;36:e00039-23.
- Turner AM, Li L, Monk IR, Lee JYH, Ingle DJ, Portelli S, et al. Rifaximin prophylaxis causes resistance to the last-resort antibiotic daptomycin. Nature. 2024 Nov;635:969-77.
- Boll L, Kern WV, Schuster S, Schultheiß M, Schneider C, Vavra M, et al. Frequent high-level rifaximin resistance in Escherichia coli associated with long-term treatment of patients with liver cirrhosis: a prospective, controlled study. Microbiol Spectr. 2025;13:e0334724.
- Pimentel M, Gunsalus RP, Rao SSC, Zhang H. Methanogens in Human Health and Disease. The American Journal of Gastroenterology Supplements. 2012;1:28-33.
- Triantafyllou K, Chang C, Pimentel M. Methanogens, Methane and Gastrointestinal Motility. J Neurogastroenterol Motil. 2014;20:31-40.
- Levitt MD, Furne JK, Kuskowski M, Ruddy J. Stability of Human Methanogenic Flora Over 35 Years and a Review of Insights Obtained From Breath Methane Measurements. Clinical Gastroenterology and Hepatology. 2006;4:123-9.
- Florin THJ, Zhu G, Kirk KM, Martin NG. Shared and unique environmental factors determine the ecology of methanogens in humans and rats. The American Journal of Gastroenterology. 2000;95:2872-9.
- Kumpitsch C, Fischmeister FPhS, Mahnert A, Lackner S, Wilding M, Sturm C, et al. Reduced B12 uptake and increased gastrointestinal formate are associated with archaeome-mediated breath methane emission in humans. Microbiome. 2021;9:193.
- Hansen EE, Lozupone CA, Rey FE, Wu M, Guruge JL, Narra A, et al. Pan-genome of the dominant human gut-associated archaeon, Methanobrevibacter smithii, studied in twins. Proc Natl Acad Sci U S A. 2011;108(Suppl 1):4599-606.
- Hoegenauer C, Hammer HF, Mahnert A, Moissl-Eichinger C. Methanogenic archaea in the human gastrointestinal tract. Nat Rev Gastroenterol Hepatol. 2022;19:805-13.
- Min YW, Rezaie A, Pimentel M. Bile Acid and Gut Microbiota in Irritable Bowel Syndrome. J Neurogastroenterol Motil. 2022;28:549-61.
- An C, Chon H, Ku W, Eom S, Seok M, Kim S, et al. Bile Acids: Major Regulator of the Gut Microbiome. Microorganisms. 2022;10:1792.
- Florin TH, Jabbar IA. A possible role for bile acid in the control of methanogenesis and the accumulation of hydrogen gas in the human colon. J Gastroenterol Hepatol. 1994;9:112-7.
- Djemai K, Drancourt M, Tidjani Alou M. Bacteria and Methanogens in the Human Microbiome: a Review of Syntrophic Interactions. Microb Ecol. 2022;83:536-54.
- Fiedorek SC, Pumphrey CL, Casteel HB. Breath methane production in children with constipation and encopresis. J Pediatr Gastroenterol Nutr. 1990;10:473-7.
- Pimentel M, Mayer AG, Park S, Chow EJ, Hasan A, Kong Y. Methane production during lactulose breath test is associated with gastrointestinal disease presentation. Dig Dis Sci. 2003;
48:86-92. - Chatterjee S, Park S, Low K, Kong Y, Pimentel M. The Degree of Breath Methane Production in IBS Correlates With the Severity of Constipation. Official journal of the American College of Gastroenterology | ACG. 2007;102:837.
- Mehravar S, Takakura W, Wang J, Pimentel M, Nasser J, Rezaie A. Symptom Profile of Patients With Intestinal Methanogen Overgrowth: A Systematic Review and Meta-analysis. Clinical Gastroenterology and Hepatology. 2025;23:1111-1122.e9.
- Pimentel M, Lin HC, Enayati P, van den Burg B, Lee HR, Chen JH, et al. Methane, a gas produced by enteric bacteria, slows intestinal transit and augments small intestinal contractile activity. American Journal of Physiology-Gastrointestinal and Liver Physiology. 2006;290:G1089-95.
- Jahng J, Jung IS, Choi EJ, Conklin JL, Park H. The effects of methane and hydrogen gases produced by enteric bacteria on ileal motility and colonic transit time. Neurogastroenterology & Motility. 2012;24:185-e92.
- Attaluri A, Jackson M, Valestin J, Rao SS. Methanogenic Flora Is Associated With Altered Colonic Transit but Not Stool Cha
racteristics in Constipation Without IBS. Official journal of the American College of Gastroenterology | ACG. 2010;105:1407. - Talamantes S, Steiner F, Spencer S, Neshatian L, Sonu I. Intestinal Methanogen Overgrowth (IMO) Is Associated with Delayed Small Bowel and Colonic Transit Time (TT) on the Wireless Motility Capsule (WMC). Dig Dis Sci. 2024;69:3361-8.
- Procházková N, Falony G, Dragsted LO, Licht TR, Raes J, Roager HM. Advancing human gut microbiota research by considering gut transit time. Gut. 2023 Jan;72:180-91.
- Tottey W, Feria-Gervasio D, Gaci N, Laillet B, Pujos E, Martin JF, et al. Colonic Transit Time Is a Driven Force of the Gut Microbiota Composition and Metabolism: In Vitro Evidence. J Neurogastroenterol Motil. 2017;23:124-34.
- El Oufir L, Flourié B, Bruley des Varannes S, Barry JL, Cloarec D, Bornet F, et al. Relations between transit time, fermentation products, and hydrogen consuming flora in healthy humans. Gut. 1996;38:870-7.
- Kim G, Deepinder F, Morales W, Hwang L, Weitsman S, Chang C, et al. Methanobrevibacter smithii Is the Predominant Methanogen in Patients with Constipation-Predominant IBS and Methane on Breath. Dig Dis Sci. 2012;57:3213-8.
- Di Stefano M, Mengoli C, Bergonzi M, Klersy C, Pagani E, Miceli E, et al. Breath Methane Excretion Is not An Accurate Marker of Colonic Methane Production in Irritable Bowel Syndrome. Official journal of the American College of Gastroenterology | ACG. 2015;110:891.
- Pimentel M, Constantino T, Kong Y, Bajwa M, Rezaei A, Park S. A 14-Day Elemental Diet Is Highly Effective in Normalizing the Lactulose Breath Test. Dig Dis Sci. 2004;49:73-7.
- O’Dwyer D. Homemade Elemental Diet to Treat Intestinal Methanogen Overgrowth: A Case Report. Integr Med (Encinitas). 2021;20:32–41.
- Eetemadi A, Tagkopoulos I. Methane and fatty acid metabolism pathways are predictive of Low-FODMAP diet efficacy for patients with irritable bowel syndrome. Clin Nutr. 2021;40:4414-21.
- Dridi B, Fardeau ML, Ollivier B, Raoult D, Drancourt M. The antimicrobial resistance pattern of cultured human methanogens reflects the unique phylogenetic position of archaea. J Antimicrob Chemother. 2011;66:2038-44.
- Pimentel M, Chatterjee S, Chow EJ, Park S, Kong Y. Neomycin Improves Constipation-Predominant Irritable Bowel Syndrome in a Fashion That Is Dependent on the Presence of Methane Gas: Subanalysis of a Double-Blind Randomized Controlled Study. Dig Dis Sci. 2006;51:1297-301.
- Ghoshal UC, Srivastava D, Verma A, Misra A. Slow Transit Constipation Associated With Excess Methane Production and Its Improvement Following Rifaximin Therapy: A Case Report. J Neurogastroenterol Motil. 2011;17:185-8.
- Ojetti V, Ianiro G, Tortora A, D’Angelo G, Di Rienzo TA, Bibbò S, et al. The effect of Lactobacillus reuteri supplementation in adults with chronic functional constipation: a randomized, double-blind, placebo-controlled trial. J Gastrointestin Liver Dis. 2014;23:387-91.
- Ojetti V, Petruzziello C, Migneco A, Gnarra M, Gasbarrini A, Franceschi F. Effect of Lactobacillus reuteri (DSM 17938) on methane production in patients affected by functional constipation: a retrospective study. Eur Rev Med Pharmacol Sci. 2017;21.
- Gottlieb K, Wacher V, Sliman J, Pimentel M. Review article: inhibition of methanogenic archaea by statins as a targeted management strategy for constipation and related disorders. Alimentary Pharmacology & Therapeutics. 2016;43:197-212.
- Demonfort Nkamga V, Armstrong N, Drancourt M. In vitro susceptibility of cultured human methanogens to lovastatin. International Journal of Antimicrobial Agents. 2017;49:176-82.
- Scheithauer TPM, Dallinga-Thie GM, De Vos WM, Nieuwdorp M, Van Raalte DH. Causality of small and large intestinal microbiota in weight regulation and insulin resistance. Molecular Metabolism. 2016;5:759-70.
- Kuang J, Zheng X, Jia W. Investigating regional-specific gut microbial distribution: an uncharted territory in disease therapeutics. protein cell. 2025;16:623-40.
- Booijink CCGM, Zoetendal EG, Kleerebezem M, de Vos WM. Microbial communities in the human small intestine: coupling diversity to metagenomics. Future Microbiol. 2007;2:285-95.
- Zheng L, Kelly CJ, Colgan SP. Physiologic hypoxia and oxygen homeostasis in the healthy intestine. A Review in the Theme: Cellular Responses to Hypoxia. Am J Physiol Cell Physiol. 2015;309:
C350-60. - He G, Shankar RA, Chzhan M, Samouilov A, Kuppusamy P, Zweier JL. Noninvasive measurement of anatomic structure and intraluminal oxygenation in the gastrointestinal tract of living mice with spatial and spectral EPR imaging. Proceedings of the National Academy of Sciences. 1999;96:4586-91.
- Fisher EM, Khan M, Salisbury R, Kuppusamy P. Noninvasive monitoring of small intestinal oxygen in a rat model of chronic mesenteric ischemia. Cell Biochem Biophys. 2013;67:10.1007/s12013-013-9611-y.
- Konjar Š, Pavšič M, Veldhoen M. Regulation of Oxygen Homeostasis at the Intestinal Epithelial Barrier Site. Int J Mol Sci. 2021;22:9170.
- Albenberg L, Esipova TV, Judge CP, Bittinger K, Chen J, Laughlin A, et al. Correlation Between Intraluminal Oxygen Gradient and Radial Partitioning of Intestinal Microbiota. Gastroenterology. 2014;147:1055-1063.e8.
- Singhal R, Shah YM. Oxygen battle in the gut: Hypoxia and hypoxia-inducible factors in metabolic and inflammatory responses in the intestine. J Biol Chem. 2020;295:10493-505.
- Villanueva-Millan MJ, Leite G, Mathur R, Rezaie A, Fajardo CM, de Freitas Germano J, et al. Hydrogen Sulfide and Methane on Breath Test Correlate with Human Small Intestinal Hydrogen Sulfide Producers and Methanogens. Dig Dis Sci. 2025 [epub ahead of print].
- Jensen BB, Jørgensen H. Effect of dietary fiber on microbial activity and microbial gas production in various regions of the gastrointestinal tract of pigs. Applied and Environmental Microbiology. 1994;60:1897-904.
- Yilmaz B, Macpherson AJ. Delving the depths of “terra incognita” in the human intestine – the small intestinal microbiota. Nat Rev Gastroenterol Hepatol. 2025;22:71-81.
- Zhu W, Zhang X, Wang D, Yao Q, Ma GL, Fan X. Simulator of the Human Intestinal Microbial Ecosystem (SHIME®): Current Developments, Applications, and Future Prospects. Pharmaceuticals (Basel). 2024;17:1639.
- Venema K, van den Abbeele P. Experimental models of the gut microbiome. Best Pract Res Clin Gastroenterol. 2013;27:115-26.
- Rossi R, Mainardi E. Prebiotics and Probiotics Supplementation in Pigs as a Model for Human Gut Health and Disease. Biomolecules. 2025;15:665.
- Wang M, Donovan SM. Human Microbiota-Associated Swine: Current Progress and Future Opportunities. ILAR Journal. 2015;56:63-73.
- van Trijp MP, Wilms E, Ríos-Morales M, Masclee AA, Brummer RJ, Witteman BJ, et al. Using naso- and oro-intestinal catheters in physiological research for intestinal delivery and sampling in vivo: practical and technical aspects to be considered. The American Journal of Clinical Nutrition. 2021;114:843-61.
- Marquez-Ortiz RA, Leon M, Abril D, Escobar-Perez J, Florez-Sarmiento C, Parra-Izquierdo V, et al. Colonoscopy aspiration lavages for mucosal metataxonomic profiling of spondylarthritis-associated gastrointestinal tract alterations. Sci Rep. 2023;13:7015.
- Zoetendal EG, Raes J, van den Bogert B, Arumugam M, Booijink CC, Troost FJ, et al. The human small intestinal microbiota is driven by rapid uptake and conversion of simple carbohydrates. ISME J. 2012;6:1415-26.
- Nejati S, Wang J, Sedaghat S, Balog NK, Long AM, Rivera UH, et al. Smart Capsule for Targeted Proximal Colon Microbiome Sampling. Acta Biomater. 2022;154:83-96.
- Rehan M, Al-Bahadly I, Thomas DG, Young W, Cheng LK, Avci E. Smart capsules for sensing and sampling the gut: status, challenges and prospects. Gut. 2024;73:186-202.
- Stine JM, Ruland KL, Beardslee LA, Levy JA, Abianeh H, Botasini S, et al. Miniaturized Capsule System Toward Real-Time Electrochemical Detection of H2S in the Gastrointestinal Tract. Adv Healthc Mater. 2024;13:2302897.
- Berean KJ, Ha N, Ou JZ, Chrimes AF, Grando D, Yao CK, et al. The safety and sensitivity of a telemetric capsule to monitor gastrointestinal hydrogen production in vivo in healthy subjects: a pilot trial comparison to concurrent breath analysis. Aliment Pharmacol Ther. 2018;48:646-54.
- Shalon D, Culver RN, Grembi JA, Folz J, Treit PV, Shi H, et al. Profiling the human intestinal environment under physiological conditions. Nature. 2023;617:581-91.
Correspondence: Eamonn M M Quigley
Email: equigley@houstonmethodist.org
Acta Gastroenterol Latinoam 2025;55(4):276-291
 Revista ACTA Órgano Oficial de SAGE
Revista ACTA Órgano Oficial de SAGE



